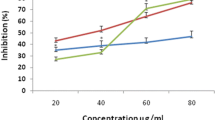
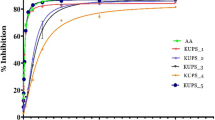
Abstract
Hedychium coronarium Koen. is considered as an endemic medicinal plant. In the current research antioxidant potential of six different extracts obtained by the successive solvent extraction method from the unexplored H. coronarium rhizome was evaluated. Various in vitro assays were performed to achieve the aim and variation in scavenging potential of the six extracts, in addition to quantification of phenolic and flavonoid compounds was done by HPLC. Among the six extracts, methanolic extract have showed highest Total phenolic content (TPC), metal chelation activity and free radical scavenging potential against DPPH, ABTS and superoxide anions as compared to other extracts. The highest scavenging potential against nitric oxide and hypochlorus acid was shown by acetone extract. The variation in anti-oxidant activity of the extracts may be due to the phyto-constituents present in different solvents. A significant correlation (r2 = 0.864) was established between antioxidant and TPC of H. coronarium from principle component analysis. Thus the present study provides strong evidence that rhizome extract of H. coronarium is a potential source of bioactive compounds and can be used as a remedy for diseases caused due to oxidative stress. Reported results could be helpful to develop novel drugs for the management of oxidative stress and associated diseases.

Similar content being viewed by others
References
Aadil KR, Barapatre A, Sudha Sahu, Harit Jha, Tiwary BN (2014) Free radical scavenging activity and reducing power of Acacia nilotica wood lignin. Int J Biol Macromol 67:220–227
Abdille MH, Singh RP, Jayaprakasha GK, Jena BS (2005) Antioxidant activity of the extracts from Dillenia indica fruits. Food Chem 90:891–896
Barapatre A, Meena AS, Mekala S, Das A, Jha H (2016) In vitro evaluation of antioxidant and cytotoxic activities of lignin fractions extracted from Acacia nilotica. Int J Biol Macromol 86:443–453
Bera TK, Chatterjee K, Ghosh D (2015) In-vitro antioxidant properties of the hydro-methanol extract of the seeds of Swietenia mahagoni (L.) Jacq. Biomark Genomic Med 7:18–24
Bharti SK, Kumar A, Krishnan S, Gupta AK, Kumar A (2015) Mechanism-based anti-diabetic activity of Fructo- and isomalto-oligosaccharides: validation by in vivo, in silico and in vitro interaction potential. Process Biochem 50:317–327
Elmastaş M, Gülçin İ, Işildak Ö, Küfrevioğlu Öİ, İbaoğlu K, Aboul-Enein HY (2006) Radical scavenging activity and antioxidant capacity of bay leaf extracts. J Iran Chem Soc 3:258–266
Gan RY, Xu XR, Song FL, Kuang L, Li HB (2010) Antioxidant activity and total phenolic content of medicinal plants associated with prevention and treatment of cardiovascular and cerebrovascular diseases. J Med Plants Res 4:2438–2444
Gonc S, Gomes D, Costa P, Romano A (2013) The phenolic content and antioxidant activity of infusions from Mediterranean medicinal plants. Ind Crops Prod 43:465–471
Hazra B, Biswas S, Mandal N (2008) Antioxidant and free radical scavenging activity of Spondias pinnata. BMC Complement Altern Med 8:1–10
Ho J (2011) Antimicrobial, mosquito larvicidal and antioxidant properties of the leaf and rhizome of Hedychium coronarium. J Chin Chem Soc 58:563–567
Hopps E, Noto D, Caimi G, Averna MR (2010) A novel component of the metabolic syndrome: the oxidative stress. NMCD 20:72–77
Iqbal E, Abu K, Lim LBL (2015) Phytochemical screening, total phenolics and antioxidant activities of bark and leaf extracts of Goniothalamus velutinus (Airy Shaw) from Brunei Darussalam. J King Saud Univ 27:224–232
Kiem PV, Thi N, Thuy K, Le H, Anh T, Xuan N, Ho Y (2011) Chemical constituents of the rhizomes of Hedychium coronarium and their inhibitory effect on the pro-inflammatory cytokines production LPS-stimulated in bone marrow-derived dendritic cells. Bioorg Med Chem Lett 21:7460–7465
Li X (2013) Solvent effects and improvements in the deoxyribose degradation assay for hydroxyl radical-scavenging. Food Chem 141:2083–2088
Li D, Jin Z, Zhou Q, Chen J, Lei Y, Sun S (2010) Discrimination of five species of Fritillaria and its extracts by FT-IR and 2D-IR. J Mol Struct 974:68–72
Liu S, Huang H (2015) Assessments of antioxidant effect of black tea extract and its rationals by erythrocyte haemolysis assay, plasma oxidation assay and cellular antioxidant activity assay. J Funct Foods 18:1095–1105
Liu A, Li L, Xu M, Lin Y, Guo H, Guo D (2006) Simultaneous quantification of six major phenolic acids in the roots of Salvia miltiorrhiza and four related traditional Chinese medicinal preparations by HPLC–DAD method. J Pharma Biomed Anal 41:48–56
Loo AY, Jain K, Darah I (2008) Antioxidant activity of compounds isolated from the pyroligneous acid, Rhizophora apiculata. Food Chem 107:1151–1160
Lu Y, Zhong CX, Wang L, Lu C, Li XL, Wang PJ (2009) Anti-inflammation activity and chemical composition of flower essential oil from Hedychium coronarium. Afr J Biotech 8:373–5377
Mesbah MK, Khalifa SI, El-Gindy A, Tawfik KA (2005) HPLC determination of certain flavonoids and terpene lactones in selected Ginkgo biloba L. phyto-pharmaceuticals. Farmaco 60:583–590
Pisoschi AM, Pop A (2015) The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem 97:55–74
Rababah TM, Ereifej KI, Esoh RB, Al-u’datt MH, Alrababah MA, Yang W (2011) Antioxidant activities, total phenolics and HPLC analyses of the phenolic compounds of extracts from common Mediterranean plants. Nat Prod Res 25:596–605
Rice-Evans CA, Miler NJ, Paganga G (1996) Structure antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Bio Med 20:933–956
Rohaeti E, Rafi M, Dyah U, Heryanto R (2015) Fourier transform infrared spectroscopy combined with chemometrics for discrimination of Curcuma longa, Curcuma xanthorrhiza and Zingiber cassumunar. Spectrochim Acta Part A Mol Biomol Spectrosc 137:1244–1249
Shahwar D, Raza MA (2012) Antioxidant potential of phenolic extracts of Mimusops elengi. APJTB 2:547–550
Siddhuraju P, Becker K (2003) Antioxidant properties of various solvent extracts of total phenolic constituents from three different agro climatic regions of drum-stick tree (Moringa olifera Lam.) leaves. J Agric Food Chem 51:2144–2155
Singh R, Singh S, Kumar S, Arora S (2007) Evaluation of antioxidant potential of ethyl acetate extract/fractions of Acacia auriculiformis A. Cunn. Food Chem Toxicol 45:1216–1223
Starlin T, Arul C, Ragavendran P, Gopalakrishnan VK (2012) Phytochemical screening, functional groups and element analysis of tylophora pauciflora wight and arn. Int Res J Pharm 3:180–183
Suresh G, Reddy PP, Babu KS, Shaik TB, Vardhan KS (2010) Two new cytotoxic labdane diterpenes from the rhizomes of Hedychium coronarium. Bioorg Med Chem Lett 20:7544–7548
Visioli F, Bellomo G, Galli C (1998) Free radical-scavenging properties of olive oil polyphenols. Biochem Biophys Res Comm 247:60–64
Wang H, Gao XD, Zhou GC, Cai L, Yao WB (2008) In vitro and in vivo antioxidant activity of aqueous extract from Choerospondias axillaris fruit. Food Chem 106:888–895
Wong JY, Matanjun P, Houi Ooi YB, Chia KF (2014) Evaluation of antioxidant activities in relation to total phenolics and flavonoids content of selected malaysian wild edible plants by multivariate analysis. Int J Food Prop 17:1763–1778
Yen GC, Duh PD, Su HJ (2005) Antioxidant properties of lotus seed and its effect on DNA damage in human lymphocytes. Food Chem 89:379–385
Zhao X, Zhu H, Chen J, Ao Q (2015) FTIR, XRD and SEM analysis of ginger powders with different size. J Food Process Preserv 39:1–10
Acknowledgements
Authors are thankful to the Head, Department of Biotechnology, GGU, Bilaspur (India) for his kind support and providing the laboratory facility. Authors are thankful to SCIT, Cochin, India and National Institute of Technology (NIT), Raipur for FTIR analysis and NIPER, Mohali for HPLC analysis. The Authors acknowledge the help and suggestion of Anand Barapatre during experiments. Financial assistance to SKP as JRF for Ph.D from University Grants Commission (UGC), New Delhi, India is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Panigrahy, S.K., Kumar, A. & Bhatt, R. Antioxidant potentials of successive solvent extracts from the unexplored Hedhychium coronarium rhizome. J Food Sci Technol 54, 3297–3306 (2017). https://doi.org/10.1007/s13197-017-2777-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-017-2777-3