Abstract
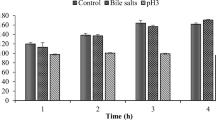
A total of 38 lactic acid bacteria, belonging to Lactobacillus, isolated from 24 samples of traditional Egyptian dairy products, were screened for antimicrobial activity against different Gram-positive and Gram-negative bacteria. A strain of Lactobacillus brevis showed the best inhibitory activity when tested by well diffusion assay. The antibacterial activity was pronounced between early logarithmic and early stationary phases. The strain produced a heat-stable antimicrobial compound showing no reduction in activity after heat treatment from 60 to 100°C for 15 and 30 min. Since it was inactivated by proteolytic enzymes, it is considered to be proteinaceous in nature and, therefore, referred to as a bacteriocin-like substance. This compound was also active over a wide pH range (pH 2–6). The antimicrobial compound was partially purified by 40% ammonium sulfate precipitation. Lactobacillus brevis was tested for its in vitro antibiotics susceptibility, tolerance to bile salts, resistance to low pH values, acidifying activity, proteolytic activity, and haemolytic activity. The results showed the potential of L. brevis strain as a probiotic culture, and hence it can be utilized in the manufacturing of pharmaceuticals and dietary supplements.


Similar content being viewed by others
References
Abdel-Bar N, Harris ND, Rill RL (1987) Purification and properties of an antimicrobial substance produced by Lactobacillus bulgaricus. J Food Sci 52:411–415
Abo-Amer AE (2007) Characterization of a bacteriocin-like inhibitory substance produced by Lactobacillus plantarum isolated from Egyptian home-made yogurt. ScienceAsia 33(3):313–319
Altschul SF, Thomas LM, Alejandro AS, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25(17):3389–3402
Axelsson L (1998) In: Salminen S, von Wright A (eds) Lactic acid bacteria: microbiology and functional aspects. Marcel Dekker, New York
Ayad EHE, Nashat S, El-Sadek N, Metwaly H, El-Soda M (2004) Selection of wild lactic acid bacteria isolated from traditional Egyptian dairy products according to production and technological criteria. Food Microbiol 21:715–725
Baumgartner A, Kueffer M, Simmen A, Grand M (1998) Relatedness of Lactobacillus rhamnosus strains isolated from clinical specimens and such from foodstuffs, humans and technology. Lebensm Wiss Technol 31:489–494
Brink BT, Minekus M, Vander Vossen JMB, Leer RJ, Huis JHJ (1994) Antimicrobial activity of lactobacilli: preliminary characterization and optimization of production of acidocin B., a novel bacteriocin produced by L. acidophilus M46. J Appl Bacteriol 77:140–148
Carr FJ, Hill D, Maida N (2002) The lactic acid bacteria: A literature survey. Crit Rev Microbiol 28:281–370
Clinical and Laboratory Standards Institute (CLSI) (2009) Performance Standards for Antimicrobial Disk Susceptibility Tests; Approved Standard (10th Ed). CLSI document M02 A10- Clinical and Laboratory Standards Institute, Wayne, PA
Coppola R, Succi M, Tremonte P, Reale A, Salzano G, Sorrentino E (2005) Antibiotic susceptibility of Lactobacillus rhamnosus strains isolated from Parmigiano Reggiano cheese. Lait 85:193–204
Daba H, Pandian S, Gosselin JF, Simard RE, Huang J, Lacroix C (1991) Detection and activity of bacteriocin produced by Leuconostoc mesenteriodes. Appl Environ Microbiol 57:3450–3455
Dal Bello F, Hertel C (2006) Oral cavity as natural reservoir for intestinal lactobacilli. Syst Appl Microbiol 29:69–76
De Man JC, Rogosa M, Sharpe ME (1960) A medium for the cultivation of lactobacilli. J Appl Bacteriol 23:130–135
Delves-Broughton J, Blackburn P, Evans RJ, Hugenholtz J (1996) Applications of the bacteriocin, nisin. Antonie Leeuwenhoek 69(2):193–202
Dunne C, Murphy L, Flynn S, O’Mahony L, O’Halloran S, Feeney M, Morrissey D, Thornton G, Fitzgerald G, Daly C, Kiely B, Quigley EMM, O’Sullivan GC, Shanahan F, Kevin J (1999) Probiotics: from myth to reality. Demonstration of functionality in animal models of disease and in human clinical trials. Antonie Leeuwenhoek 76:279–292
Dunne C, O’Mahony L, Murphy L, Thornton G, Morrissey D, O’Halloran S, Feeney M, Flynn S, Fitzgerald G, Daly C, Kiely B, O’Sullivan GC, Shanahan F, Collins JK (2001) In vitro selection criteria for probiotic bacteria of human origin: correlation with in vivo findings. Am J Clin Nutr 73(suppl):386S–392S
Elegado FB, Guerra MA, Macayan RA, Mendoza HA, Lirazan MB (2004) Spectrum of bacteriocin activity of Lactobacillus plantarum BS and fingerprinting by RAPD-PCR. Int J Food Microbiol 95:11–18
El-Naggar MYM (2004) Comparative study of Probiotic cultures to control the growth of Escherichia coli O157:H7 and Salmonella tybhimurium. Biotechnol 3:173–180
Ennahar S, Zendo T, Sonomoto K, Ishizaki A (1999) Investigation of bacetriocin production and purification from Nukadoko Isolates displaying anitimicrobial activity. Jpn J Lactic Acid Bacteria 10:29–36
Ennahar S, Sashihara T, Sonomoto K, Ishizaki A (2000) Class IIa bacteriocins: biosynthesis, structure and activity. FEMS Microbiol Rev 24:85–161
Espeche MC, Otero MC, Sesma F, Nader-Macias FME (2009) Screening of surface properties and antagonistic substances production by lactic acid bacteria isolated from the mammary gland of healthy and mastitic cows. Vet Microbiol 135:346–357
Forbes BA, Sahm DF, Weissfeld AS (1998) Bailey and Scott's Diagnostic Microbiology, 10th edn. Mosby, St. Louis
Fricourt BV, Barefoot SF, Testin RF, Haysaka SS (1994) Detection and activity of plantaricin F an antimicrobial substance from Lactobacillus plantarum BF001 isolated from processed channel catfish. J Food Prot 57:698–702
Fuller R (1989) Probiotics in man and animals. J Appl Bacteriol 66:365–378
Gilliand SE (1990) Health and nutritional benefits from lactic acid bacteria. FEMS Microbiol Rev 87:175–178
Gong HS, Meng XC, Wang H (2010) Plantaricin MG active against Gram-negative bacteria produced by Lactobacillus plantarum KLDS1.0391 isolated from “Jiaoke”, a traditional fermented cream from China. Food Control 21:89–96
Halami PM, Chandrashekar A, Nand K (2000) Lactobacillus farciminis MD, a newer strain with potential for bacteriocin and antibiotic assay. Lett Appl Microbiol 30:197–202
Havenaar R, Brink BT, Huis In’T Veld JHJ (1992) Selection of strains for probiotic use. In: Fuller R (ed) Probiotics. The Scientific Basis. Chapman and Hall, London
Iñiguez-Palomares C, Pérez-Morales R, Acedo-Félix E (2007) Evaluation of probiotic properties in Lactobacillus isolated from small intestine of piglets. Rev Latinoam Microbiol 49(3–4):46–54
Jack RW, Tagg JR, Ray B (1995) Bacteriocins of Gram-positive bacteria. Microbiol Rev 59(2):171–200
Jamaly N, Benjouad A, Bouksaim M (2011) Probiotic potential of Lactobacillus strains isolated from known popular traditional moroccan dairy products. Brit Microbiol Res J 1(4):79–94
Kacem M, Karam N-E (2006) In vitro preselection criteria for probiotic Lactobacillus plantarum strains of fermented olive origin. Int J Probiotics Prebiotics 1(1):27–32
Karthikeyan V, Santosh SW (2009) Isolation and partial characterization of bacteriocin produced from Lactobacillus plantarum. Afr J Microbiol Res 3(5):233–239
Kelly WJ, Asmundson RV, Huang CM (1996) Characterization of plantaricin KW30, a bacteriocin produced by Lactobacillus plantarum. J Appl Bacteriol 81:657–662
Khay E, Idaomar M, Castro LMP, Bernárdez PF, Senhaji NS, Abrini J (2011) Antimicrobial activities of the bacteriocin-like substances produced by lactic acid bacteria isolated from Moroccan dromedary milk. Afr J Biotechnol 10(51):10447–10455
Klaenhammer TR (1993) Genetics of bacteriocins produced by lactic acid bacteria. FEMS Microbiol Rev 12:39–85
Lee YK, Salminen S (1995) The coming age of probiotics. Trends Food Sci Technol 6:241–245
Maragkoudakis PA, Zoumpopoulou G, Miaris C, Kalantzopoulos G, Pot B, Tsakalidou E (2006) Probiotic potential of Lactobacillus strains isolated from dairy products. Int Dairy J 16:189–199
Marco ML, Pavan S, Kleerebezem M (2006) Towards understanding molecular modes of probiotic action. Curr Opin Biotechnol 17:204–210
McFarland LV, Dublin S (2008) Meta-analysis of probiotics for the treatment of irritable bowel syndrome. World J Gastroenterol 14:2650–2661
Mishra V, Prasad D (2005) Application of in vitro methods for selection of Lactobacillus casei strains as potential probiotics. Int J Food Microbiol 103:109–115
Morata de Ambrosini V, González S, De Ruiz Holgado AP, Oliver G (1998) Study of the morphology of the cell walls of some strains of lactic acid bacteria and related species. J Food Prot 61:557–562
Morisset D, Berjeaud JM, Marion D, Lacombe C, Frère J (2004) Mutational analysis of mesentericin Y105, an anti-Listeria bacteriocin, for determination of impact on bactericidal activity, in vitro secondary structure, and Membrane Interaction. Appl Environ Microbiol 70:4672–4680
Nelson RRS (1999) Intrinsically vancomycin-resistant Gram-positive organisms: clinical relevance and implications for infection control. J Hosp Infect 42:275–282
Nettles CG, Barefoot SF (1993) Biochemical and genetic characteristics of bacteriocins of food-associated lactic acid bacteria. J Food Prot 56:338–356
Nowroozi J, Mirzaii M, Norouzi M (2004) Study of Lactobacillus as Probiotic Bacteria. Iran J Public Health 33(2):1–7
Ogunbanwo ST, Sanni AI, Onilude AA (2003a) Characterization of bacteriocin produced by Lactobacillus plantarum F1 and Lactobacillus brevis OG1. Afr J Biotechnol 2:219–227
Ogunbanwo ST, Sanni AI, Onilude AA (2003b) Influence of cultural conditions on the production of bacteriocin by Lactobacillus brevis OG1. Afr J Biotechnol 2(7):179–184
Ogunshe AAO, Omotoso MA, Adeyeye JA (2007) In vitro antimicrobial characteristics of bacteriocin producing Lactobacillus strains from Nigerian indigenous fermented foods. Afr J Biotechnol 6(4):445–453
Oh S, Kim SH, Worobo RW (2000) Characterization and purification of a bacteriocin produced by a potential probiotic culture, Lactobacillus acidophilus 30SC. J Dairy Sci 83:2747–2752
Olukoya DK, Tichazek PS, Butsch A, Vogel RF, Hammes WP (1993) Charaterization of the bacteriocins produced by Lactococcus pentosus DK7 isolated from ogi and Lactococcus plantarum DK9 from fufu. Chem Mikrobiol Technolo Lebensmittel 15:65–68
Pham M, Lemberg DA, Day AS (2008) Probiotics: sorting the evidence from the myths. Med J Aust 188:304–308
Piard JC, Delorme F, Giraffa G, Commissaire J, Desmazeaud M (1990) Evidence for a bacteriocin produced by Lactococcus lactis CNRZ 481. Neth Milk Dairy J 44:143–158
Ponce AG, Moreira MR, del Valle CE, Roura SI (2008) Preliminary characterization of bacteriocin-like substances from lactic acid bacteria isolated from organic leafy vegetables. LWT - Food Sci Technol 41:432–441
Rochelle PA, Will JAK, Fry JC, Jenkins GJS, Parkes RJ, Turley CM, Weightman AJ (1995) In: Trevors JT, van Elsas JD (eds) Nucleic Acids in the Environment. Springer, Berlin
Salminen S, Wright A, Morelli L, Marteau P, Brassart D, de Vos WM, Fonden R, Saxelin M, Collins K, Mogensen G, Birkeland SE, Mattila-Sandholm T (1998) Demonstration of safety of probiotics, a review. Int J Food Microbiol 44:93–106
Sambrook J, Fritsch EF, Maniatis T (2001) Molecular Cloning: a Laboratory Manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Santini C, Baffoni L, Gaggia F, Granata M, Gasbarri R, Diana Di Gioia D, Biavati B (2010) Characterization of probiotic strains: An application as feed additives in poultry against Campylobacter jejuni. Int J Food Microbiol 141:S98–S108
Schillinger U, Guigas C, Holzapfel WH (2005) In vitro adherence and other properties of lactobacilli used in probiotic yoghurt-like products. Int Dairy J 12:1289–1297
Schnürer J, Magnusson J (2005) Antifungal lactic acid bacteria as biopreservatives. Trends Food Sci Technol 16:70–78
Sit CS, Vederas JC (2008) Approaches to the discovery of new antibacterial 342 agents based on bacteriocins. Biochem Cell Biol 86:116–123
Soomro AH, Masud T, Anwaar K (2002) Role of lactic acid bacteria (LAB) in food preservation and human health - a review. Pak J Nutr 1(1):20–24
Stiles ME (1996) Biopreservation by lactic acid bacteria. Antonie Leeuwenhoek 70:331–345
Todorov S, Onno B, Sorokine O, Chobert JM, Ivanova I, Dousset X (1999) Detection and characterization of a novel antibacterial substance produced by Lactobacillus plantarum ST 31 isolated from sourdough. Int J Food Microbiol 48:167–177
Todorov S, Nyati H, Meincken M, Dicks LMT (2007) Partial characterization of bacteriocin AMA-K, produced by Lactobacillus plantarum AMA-K isolated from naturally fermented milk from Zimbabwe. Food Control 18(6):656–664
Vaughan A, Eijsink VGH, O' Sullivan EF, O' Hanlon K, Van Sinderen D (2001) An analysis of bacteriocins produced by lactic acid bacteria isolated from malted barley. J Appl Microbiol 91:131–138
Verellen TLJ, Van Reenen GBCA, Dicks LMT, Vandamme EJ (1998) Fermentation optimization of plantaricin 423, a bacteriocin produced by Lactobacillus plantarum 423. J Ferment Bioeng 86:174–179
Acknowledgement
I warmly thank Dr. Shaimaa Sobieh Lecturer in Cytology and Genetics, University College for Women (Arts, Science and Education), Ain Shams University, for her supportive attitude towards our work, and for valuable comments and review on the part of identification.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rushdy, A.A., Gomaa, E.Z. Antimicrobial compounds produced by probiotic Lactobacillus brevis isolated from dairy products. Ann Microbiol 63, 81–90 (2013). https://doi.org/10.1007/s13213-012-0447-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13213-012-0447-2