Abstract
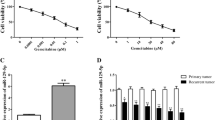
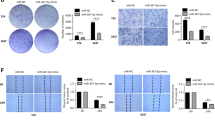
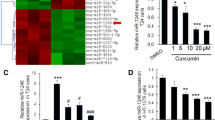
Arsenic trioxide has shown remarkable biological activity against bladder cancer in some clinical studies. However, the mechanism of its action is unknown. Our aim was to find the relationship between miRNAs and arsenic trioxide treatment by using T24 human bladder carcinoma cells. By performing microRNA microarray and quantitative real-time PCR after ATO treatment, we found that expression levels of several miRNAs, in particular, miRNA-19a, were significantly decreased in T24 cell line. Furthermore, cell proliferation assay, flow cytometry analysis, prediction of miRNA targets, Western blot analysis, and luciferase reporter assay were performed to determine the role of mir-19a in affecting the biological behaviors of T24 cells. Several miRNAs were up-regulated or down-regulated in T24 cells treated with arsenic trioxide compared to their controls. If only changes above two folds were considered, two miRNAs were identified, miRNA-19a was down-regulated, while miRNA-222* was up-regulated. Among them, knockdown of miRNA-19a by anti-miRNA-19a transfection showed a positive therapeutic effect in bladder cancer cells by inhibiting cell growth and inducing cell apoptosis targeting PTEN through the PTEN/Akt pathway. Besides this, a synergy effect was detected between knockdown of miRNA-19a and arsenic trioxide. Arsenic trioxide altered miRNA expression profile in T24 cells. It seems miRNA-19a plays a critical role in the mechanism of arsenic trioxide treatment in bladder cancer. The synergy effect between miRNA-19a and arsenic trioxide that advocates targeting the mir-19a may represent a potential approach to enhance the efficacy and safety of ATO to treat bladder cancer by a decrease in dose.




Similar content being viewed by others
References
Shen ZX, Chen GQ, Ni JH, et al. Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients. Blood. 1997;89:3354–60.
Soignet SL, Frankel SR, Douer D, et al. United States multicenter study of arsenic trioxide in relapsed acute promyelocytic leukemia. J Clin Oncol. 2001;19:3852–60.
Chen X, Zhang M, Liu LX. The overexpression of multidrug resistance-associated proteins and gankyrin contribute to arsenic trioxide resistance in liver and gastric cancer cells. Oncol Rep. 2009;22:73–80.
Karlsson J, Pietras A, Beckman S, Pettersson HM, Larsson C, Pahlman S. Arsenic trioxide-induced neuroblastoma cell death is accompanied by proteolytic activation of nuclear Bax. Oncogene. 2007;26:6150–9.
Lin TH, Kuo HC, Chou FP, Lu FJ. Berberine enhances inhibition of glioma tumor cell migration and invasiveness mediated by arsenic trioxide. BMC Cancer. 2008;8:58.
Murgo AJ. Clinical trials of arsenic trioxide in hematologic and solid tumors: overview of the National Cancer Institute Cooperative Research and Development Studies. Oncologist. 2001;6 Suppl 2:22–8.
Bi X, Gu J, Guo Z, et al. Different pathways are involved in arsenic-trioxide-induced cell proliferation and growth inhibition in human keratinocytes. Skin Pharmacol Physiol. 2010;23:68–78.
Cai BZ, Meng FY, Zhu SL, et al. Arsenic trioxide induces the apoptosis in bone marrow mesenchymal stem cells by intracellular calcium signal and caspase-3 pathways. Toxicol Lett. 2010;193:173–8.
Han YH, Moon HJ, You BR, Kim SZ, Kim SH, Park WH. Effects of arsenic trioxide on cell death, reactive oxygen species and glutathione levels in different cell types. Int J Mol Med. 2010;25:121–8.
Han YH, Moon HJ, You BR, Kim SZ, Kim SH, Park WH. The effect of MAPK inhibitors on arsenic trioxide-treated Calu-6 lung cells in relation to cell death, ROS and GSH levels. Anticancer Res. 2009;29:3837–44.
Mandegary A, Hosseini R, Ghaffari SH, et al. The expression of p38, ERK1 and Bax proteins has increased during the treatment of newly diagnosed acute promyelocytic leukemia with arsenic trioxide. Ann Oncol. 2010;21:1884–90.
Mann KK, Colombo M, Miller Jr WH. Arsenic trioxide decreases AKT protein in a caspase-dependent manner. Mol Cancer Ther. 2008;7:1680–7.
Wen J, Feng Y, Huang W, et al. Enhanced antimyeloma cytotoxicity by the combination of arsenic trioxide and bortezomib is further potentiated by p38 MAPK inhibition. Leuk Res. 2010;34:85–92.
Friedland DR, Eernisse R, Erbe C, Gupta N, Cioffi JA. Cholesteatoma growth and proliferation: posttranscriptional regulation by microRNA-21. Otol Neurotol. 2009;30:998–1005.
Guo L, Ding ZH. Advances in the studies of miRNAs and cell apoptosis. Sheng Li Ke Xue Jin Zhan. 2007;38:331–5.
Li Y, Guessous F, Zhang Y, et al. MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes. Cancer Res. 2009;69:7569–76.
Vecchione A, Croce CM. Apoptomirs: small molecules have gained the license to kill. Endocr Relat Cancer. 2010;17:F37–50.
Ren Y, Zhou X, Mei M, et al. MicroRNA-21 inhibitor sensitizes human glioblastoma cells U251 (PTEN-mutant) and LN229 (PTEN-wild type) to taxol. BMC Cancer. 2010;10:27.
Zhong M, Ma X, Sun C, Chen L. MicroRNAs reduce tumor growth and contribute to enhance cytotoxicity induced by gefitinib in non-small cell lung cancer. Chem Biol Interact. 2010;184:431–8.
Thomson JM, Parker J, Perou CM, Hammond SM. A custom microarray platform for analysis of microRNA gene expression. Nat Methods. 2004;1:47–53.
Xiao F, Zuo Z, Cai G, Kang S, Gao X, Li T. miRecords: an integrated resource for microRNA-target interactions. Nucleic Acids Res. 2009;37:D105–110.
Wang Y, An R, Dong X, Pan S, Duan G, Sun X. Protein kinase C is involved in arsenic trioxide-induced apoptosis and inhibition of proliferation in human bladder cancer cells. Urol Int. 2009;82:214–21.
Akao Y, Nakagawa Y, Hirata I, et al. Role of anti-oncomirs miR-143 and -145 in human colorectal tumors. Cancer Gene Ther. 2010;17:398–408.
Li Y, Kong D, Wang Z, Sarkar FH. Regulation of microRNAs by Natural Agents: an emerging field in chemoprevention and chemotherapy research. Pharm Res. 2010;27:1027–41.
Doebele C, Bonauer A, Fischer A, et al. Members of the microRNA-17-92 cluster exhibit a cell intrinsic anti-angiogenic function in endothelial cells. Blood. 2010;115:4631–3.
Loven J, Zinin N, Wahlstrom T, et al. MYCN-regulated microRNAs repress estrogen receptor-alpha (ESR1) expression and neuronal differentiation in human neuroblastoma. Proc Natl Acad Sci U S A. 2010;107:1553–8.
Pezzolesi MG, Platzer P, Waite KA, Eng C. Differential expression of PTEN-targeting microRNAs miR-19a and miR-21 in Cowden syndrome. Am J Hum Genet. 2008;82:1141–9.
Wang R, Li H, Guo G, et al. Augmentation by carnosic acid of apoptosis in human leukaemia cells induced by arsenic trioxide via upregulation of the tumour suppressor PTEN. J Int Med Res. 2008;36:682–90.
Lewis BP, Shih IH, Jones-Rhoades MW, Bartel DP, Burge CB. Prediction of mammalian microRNA targets. Cell. 2003;115:787–98.
Conflict of interest statement
None declared
Author information
Authors and Affiliations
Corresponding author
Additional information
Yan Cao and Shi-Liang Yu are contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cao, Y., Yu, SL., Wang, Y. et al. MicroRNA-dependent regulation of PTEN after arsenic trioxide treatment in bladder cancer cell line T24. Tumor Biol. 32, 179–188 (2011). https://doi.org/10.1007/s13277-010-0111-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-010-0111-z