Abstract
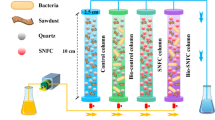
Laboratory-scale experiments were carried out to investigate the removal efficiency of iron and manganese from groundwater using limestone filter assisted with iron-oxidized bacteria. Column studies were conducted using limestone with a particle size between 0.6 and 2.36 mm and run at different operation times with a flow rate of 4 L/h and retention time of 2.94 min. Iron-oxidized bacteria were extracted from groundwater samples and cultured in an agar plate with nutrient agar solution for the bacteria duplication. Iron-oxidized bacteria resulted in a change in limestone composition as shown in X-ray diffraction and scanning electron microscope results due to the destruction of limestone particles for easier adsorption. Results show that the limestone filter assisted with IOB has better performance than the filter of limestone alone, in removing Fe and Mn. The maximum removal efficiency of Fe and Mn was 81.72% and 83.63%, respectively, at an operation time of 2 h. Iron-oxidized bacteria catalyze the oxidation of adsorption process as well as the soluble Fe(II) and Mn(II) to insoluble Fe(III) and Mn(IV). In addition, the results revealed that the increase in operation time resulted in increasing the rate of Fe and Mn oxidation. Operation time was found to be an influential factor on the removal process where the optimum values of pH and turbidity were achieved at 1.5 h of operation time. Furthermore, Fe concentration was decreased with the increase in operation time, while Mn concentration was increased with the increase in operation time. Overall, this study demonstrates that the limestone filter assisted with iron-oxidized bacteria is an effective filtration process for Fe and Mn removal in groundwater.








Similar content being viewed by others
References
Abdulkadir M, Hernandez-Perez V, Lowndes IS, Azzopardi BJ, Sam-Mbomah E (2016) Experimental study of the hydrodynamic behaviour of slug flow in a horizontal pipe. Chem Eng Sci 156:147–161. https://doi.org/10.1016/j.ces.2016.09.015
Alfarrah N, Walraevens K (2018) Groundwater overexploitation and seawater intrusion in coastal areas of arid and semi-arid regions. Water 10:143–154. https://doi.org/10.3390/w10020143
APHA (2012) Standard methods for the examination of water and wastewater 22. American Public Health Association, American Chemical Society, Association of Official Agricultural Chemists, Washington, DC
APHA (2015) Standard methods for the examination of water and sewage. American Public Health Association, American Chemical Society, Association of Official Agricultural Chemists, Washington, DC
Arp DJ, Stein LY (2003) Metabolism of inorganic N compounds by ammonia-oxidizing bacteria. Crit Rev Biochem Mol Biol 38:471–495. https://doi.org/10.1080/10409230390267446
Aziz HA, Smith PG (1992) The influence of pH and coarse media on manganese precipitation from water. Water Res 26:853–855. https://doi.org/10.1016/0043-1354(92)90017-X
Aziz HA, Othman N, Yusuff MS, Basri DRH, Ashaari FAH, Adlan MN, Perwira M (2001) Removal of copper from water using limestone filtration technique determination of mechanism of removal. Environ Int 26:359–399. https://doi.org/10.1016/S0160-4120(01)00018-6
Aziz HA, Yusoff MS, Adlan MN, Adnan NH, Alias S (2004) Physico-chemical removal of iron from semi-aerobic landfill leachate by limestone filter. Waste Manag 24:353–358. https://doi.org/10.1016/j.wasman.2003.10.006
Aziz HA, Adlan MN, Ariffin KS (2008) Heavy metals (Cd, Pb, Zn, Ni, Cu and Cr(III)) removal from water in Malaysia: post treatment by high quality limestone. Bioresour Technol 99:1578–1583. https://doi.org/10.1016/j.biortech.2007.04.007
Babel S, Kurniawan TA (2003) Low-cost adsorbents for heavy metals uptake from contaminated water: a review. J Hazard Mater 97:219–243. https://doi.org/10.1016/S0304-3894(02)00263-7
Bruins JH, Petrusevski B, Slokar YM, Kruithof JC, Kennedy MD (2015) Manganese removal from groundwater: characterization of filter media coating. Desalin Water Treat 55(7):1851–1863
Chaturvedi S, Dave PN (2012) Removal of iron for safe drinking water. Desalination 303:1–11. https://doi.org/10.1016/j.desal.2012.07.003
Clogston JD, Patri AK (2011) Zeta Potential Measurement. In: McNeil S (ed) Characterization of nanoparticles intended for drug delivery. Methods in molecular biology (methods and protocols). Humana Press, Totowa
Crini G, Lichtfouse E, Wilson LD, Morin-Crini N (2018) Adsorption-oriented processes using conventional and non-conventional adsorbents for wastewater treatment. In: Green adsorbents for pollutant removal. Springer, Cham, pp 23–71
Dashti AF, Aziz HA, Adlan MN, Ibrahim AH (2019) Calcined limestone horizontal roughing filter for treatment of palm oil mill effluent polishing pond. Int J Environ Sci Technol 16:6419–6430. https://doi.org/10.1007/s13762-019-02467-4
Devi RR, Umlong IM, Das B, Borah K, Thakur AJ, Raul PK, Banerjee S, Singh L (2014) Removal of iron and arsenic(III) from drinking water using iron oxide-coated sand and limestone. Appl Water Sci 4:175–182. https://doi.org/10.1007/s13201-013-0139-5
Dippenaar MA, Sole MD, Van Rooy JL, Du Toit GJ, Reynecke JL (2005) Determining actual LNAPL plume thickness: review and case study in a fractured aquifer. Bull Eng Geol Environ 64:347–360. https://doi.org/10.1007/s10064-005-0278-5
Dou X, Wang GC, Zhu M, Liu F, Li W, Mohan D, Pittman CU Jr (2018) Identification of Fe and Zr oxide phases in an iron–zirconium binary oxide and arsenate complexes adsorbed onto their surfaces. J Hazard Mater 353:340–347. https://doi.org/10.1016/j.jhazmat.2018.04.004
Du Z, Hu B, Shi A, Ma X, Cheng Y, Chen P, Ruan R (2012) Cultivation of a microalga Chlorella vulgaris using recycled aqueous phase nutrients from hydrothermal carbonization process. Bioresour Technol 126:354–357. https://doi.org/10.1016/j.biortech.2012.09.062
Du X, Liu G, Qu F, Li K, Shao S, Li G, Liang H (2017) Removal of iron, manganese and ammonia from groundwater using a PAC–MBR system: the anti-pollution ability, microbial population and membrane fouling. Desalination 403:97–106. https://doi.org/10.1016/j.desal.2016.03.002
Ehrlich HL (1999) Microbes as geologic agents: their role in mineral formation. Geomicrobiol J 16:135–153. https://doi.org/10.1080/014904599270659
El Araby R, Hawash S, El Diwani G (2009) Treatment of iron and manganese in simulated groundwater via ozone technology. Desalination 249:1345–1349. https://doi.org/10.1016/j.desal.2009.05.006
Fadel A, Atoui A, Lemaire BJ, Vinçon-Leite B, Slim K (2015) Environmental factors associated with phytoplankton succession in a Mediterranean reservoir with a highly fluctuating water level. Environ Monit Assess 187(10):633
Ghosh A, Chakrabarti S, Ghosh UC (2014) Fixed-bed column performance of Mn-incorporated iron(III) oxide nanoparticle agglomerates on As(III) removal from the spiked groundwater in lab bench scale. Chem Eng J 248:18–26. https://doi.org/10.1016/j.cej.2014.03.010
Granger HC, Stoddart AK, Gagnon GA (2014) Direct biofiltration for manganese removal from surface water. J Environ Eng 140(4):04014006
Hashim MA, Mukhopadhyay S, Sahu JN, Sengupta B (2011) Remediation technologies for heavy metal contaminated groundwater. J Environ Manag 92:2355–2388. https://doi.org/10.1016/j.jenvman.2011.06.009
He J, Chen JP (2014) A comprehensive review on biosorption of heavy metals by algal biomass: materials, performances, chemistry, and modeling simulation tools. Bioresour Technol 160:67–78. https://doi.org/10.1016/j.biortech.2014.01.068
Hedrich S, Schlömann M, Johnson D (2011) The iron-oxidizing proteobacteria. Microbiology 157:1551–1564. https://doi.org/10.1099/mic.0.045344-0
Homagai PL, Ghimire KN, Inoue K (2010) Adsorption behavior of heavy metals onto chemically modified sugarcane bagasse. Bioresour Technol 101:2067–2069. https://doi.org/10.1016/j.biortech.2009.11.073
Honary S, Zahir F (2013) Effect of zeta potential on the properties of nano-drug delivery systems—a review (Part 2). Trop J Pharm Res 12:265–273. https://doi.org/10.4314/tjpr.v12i2.20
Hu J, Dong H, Xu Q, Ling W, Qu J, Qiang Z (2018) Impacts of water quality on the corrosion of cast iron pipes for water distribution and proposed source water switch strategy. Water Res 129:428–435. https://doi.org/10.1016/j.watres.2017.10.065
Hussin NH, Yusoff I, Alias Y, Mohamad S, Rahim NY, Ashraf MA (2014) Ionic liquid as a medium to remove iron and other metal ions: a case study of the North Kelantan Aquifer, Malaysia. Environ Earth Sci 71:2105–2113. https://doi.org/10.1007/s12665-013-2615-5
Jusoh H, Sapari N, Azie RZR (2011) Removal of iron from groundwater by sulfide precipitation. World Acad Sci Eng Technol 5:652–658
Lenntech BV (2019) Lenntech water treatment solutions: turbidity. The Netherlands. https://www.lenntech.com/turbidity.htm. Accessed 02 Aug 2019
Liang L, Mcnabb JA, Paulk JM, Gu BH, Mccarthy JF (1993) Kinetics of Fe(II) oxygenation at low partial pressure of oxygen in the presence of natural organic matter. Environ Sci Technol 27:1864–1870. https://doi.org/10.1021/es00046a014
Ligate FJ, Mdoe JE (2015) Removal of heavy metal ions from aqueous solution using rice husks-based adsorbents. Tanzan J Sci 41:90–102
Lu H, Reddy EP, Smirniotis PG (2006) Calcium oxide-based sorbents for capture of carbon dioxide at high temperatures. Ind Eng Chem Res 45:3944–3949. https://doi.org/10.1021/ie051325x
Maji SK, Kao YH, Liao PY, Lin YJ, Liu CW (2013) Implementation of the adsorbent iron-oxide-coated natural rock (IOCNR) on synthetic As(III) and on real arsenic-bearing sample with filter. Appl Surf Sci 284:40–48. https://doi.org/10.1016/j.apsusc.2013.06.154
Malaysian Standard of Drinking Water Quality (MS2320) (2010) http://kmam.moh.gov.my/public-user/drinking-water-quality-standard.html. Access 04 Aug 2019
Meena AK, Kadirvelu K, Mishra GK, Rajagopal C, Nagar PN (2008) Adsorptive removal of heavy metals from aqueous solution by treated sawdust (Acacia arabica). J Hazard Mater 150(3):604–611. https://doi.org/10.1016/j.jhazmat.2007.05.030
Mojiri A, Aziz HA, Zaman NQ, Aziz SQ, Zahed MA (2016) Metals removal from municipal landfill leachate and wastewater using adsorbents combined with biological method. Desalin Water Treat 57:2819–2833. https://doi.org/10.1080/19443994.2014.983180
Ngah WW, Hanafiah MAKM (2008) Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: a review. Bioresour Technol 99:3935–3948. https://doi.org/10.1016/j.biortech.2007.06.011
Okafor PC, Okon PU, Daniel EF, Ebenso EE (2012) Adsorption capacity of coconut (Cocos nucifera L.) shell for lead, copper, cadmium and arsenic from aqueous solutions. Int J Electrochem Sci 7:12354–12369
Orbe P (2007) How do we culture bacterial colonies and prepare them for identification. http://www.scienceteacherprogram.org/biology/Orbe07.html. Accessed 27 July 2019
Patil DS, Chavan SM, Oubagaranadin JUK (2016) A review of technologies for manganese removal from wastewaters. J Environ Chem Eng 4:468–487. https://doi.org/10.1016/j.jece.2015.11.028
Rahman M, Sathasivam KV (2015) Heavy metal adsorption onto Kappaphycus sp. from aqueous solutions: the use of error functions for validation of isotherm and kinetics models. BioMed Res Int 126298:13. https://doi.org/10.1155/2015/126298
Rohrssen MK (2007) Siderite-magnetite oxygen isotope fractionation: a potential iron mineral biosignature
Romano CA, Zhou M, Song Y, Wysocki VH, Dohnalkova AC, Kovarik L, Tebo BM (2017) Biogenic manganese oxide nanoparticle formation by a multimeric multicopper oxidase Mnx. Nat Commun 8:746–760. https://doi.org/10.1038/s41467-017-00896-8
Rose AW, Means B, Shah PJ (2003) Methods for passive removal of manganese from acid mine drainage. In: Proceedings of the 24th West Virginia surface mine drainage task force symposium, pp 71–82
Silva AM, Cruz FLS, Lima RMF, Teixeira MC, Leao VA (2010) Manganese and limestone interactions during mine water treatment. J Hazard Mater 181:514–520. https://doi.org/10.1016/j.jhazmat.2010.05.044
Sonar N (2016) Concept of zeta potential. https://www.slideshare.net/NitinSonar1/concept-of-zeta-potential. Accessed 28 July 2019
Stumm W, Morgan JJ (1981) Aquatic chemistry. Wiley, New York
Uddin MK (2017) A review on the adsorption of heavy metals by clay minerals, with special focus on the past decade. Chem Eng J 308:438–462. https://doi.org/10.1016/j.cej.2016.09.029
Wang Y, Sikora S, Kim H, Boyer TH, Bonzongo J, Townsend TG (2013) Effects of solution chemistry on the removal reaction between calcium carbonate-based materials and Fe(II). Sci Total Environ 443:717–724. https://doi.org/10.1016/j.scitotenv.2012.11.009
Acknowledgments
This work was funded by Universiti Sains Malaysia under Research University Grant Scheme (Grant No. 1001.PAWAM.8014081) and Ministry of Higher Education Malaysia under FRGS Grant Scheme (Grant No: 203.PAWAM.6071412). Also, the authors acknowledged the Environmental Engineering Laboratory, School of Civil Engineering and Bioprocess laboratory, School of Industrial Technology, USM for their testing facilities.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial responsibility: R Saravanan.
Rights and permissions
About this article
Cite this article
Aziz, H.A., Tajarudin, H.A., Wei, T.H.L. et al. Iron and manganese removal from groundwater using limestone filter with iron-oxidized bacteria. Int. J. Environ. Sci. Technol. 17, 2667–2680 (2020). https://doi.org/10.1007/s13762-020-02681-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-020-02681-5