Abstract
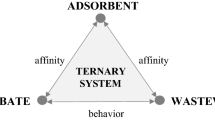
The adsorption of methyl orange(MO) on chitosan(CS) beads in aqueous solutions was investigated by a batch equilibration technique. Special emphasis was focused on the effect of sorbent concentration(c s) on the adsorption equilibration of MO on CS beads. An obvious c s-effect was observed in the adsorption equilibration, i.e., the adsorption amount(Γ) was declined with c s increase. The classical Langmuir model adequately described the adsorption isotherm for each given c s. However, it could not be used to predict the c s-effect observed. The applicability of the Langmuir-SCA isotherm, a surface component activity(SCA) model equation, to fit the c s-effect data was examined. In the SCA model, the activity coefficient of sorbent surface sites, \(f_{H_2 O}^s \), was assumed to be a function of c s due to the deviation of a real adsorption system from an ideal one, arisen from sorbent particle-particle interactions in real systems. The results show that the Langmuir-SCA isotherm could accurately describe the c s-effect observed under the studied conditions. Furthermore, the effects of temperature(t), pH, and electrolyte(NaNO3) concentration(\(c_{NaNO_3 } \)) on \(f_{H_2 O}^s \) were examined. The results show that \(f_{H_2 O}^s \) clearly decreased with increasing t(20–35 °C) and pH(5–8), but no obvious change in \(f_{H_2 O}^s \) was observed as \(c_{NaNO_3 } \) varied in a range 0.001–0.010 mol/L. These results give a better understanding of the c s-effect.
Similar content being viewed by others
References
Forgacs E., Cserhati T., Oros G., Environ. Int., 2004, 30(7), 953
Crini G., Badot P. M., Prog. Polym. Sci., 2008, 33(4), 399
Bhatnagar A., Sillanpää M., Adv. Colloid Interface Sci., 2009, 152(1), 26
Nourouzi M. M., Chuah T. G., Choong T. S. Y., Water Sci. Technol., 2011, 63(5), 984
Sadri Moghaddam S., Alavi Moghaddam M. R., Arami M., J. Hazard. Mater., 2010, 175(1), 651
Riera-Torres M., Gutiérrez-Bouzán C., Crespi M., Desalination, 2010, 252(1), 53
Koyuncu I., J. Chem. Technol. Biotechnol., 2003, 78(12), 1219
Ghoneim M. M., El-Desoky H. S., Zidan N. M., Desalination, 2011, 274(1), 22
Zanjanchi F., Hadipour N. L., Sabzyan H., Beheshtian J., Dyes and Pigments, 2011, 89(1), 16
Yavuz Y., Savaş K. A., Öğütveren Ü. B., J. Chem. Technol. Biotechnol., 2011, 86(2), 261
Wu F. C., Tseng R. L., Juang R. S., J. Hazard. Mater., 2000, 73(1), 63
Chiou M. S., Li H. Y., J. Hazard. Mater., 2002, 93(2), 233
Wen Y. Z., Liu W. Q., Fang Z. H., Liu W. P., J. Environ. Sci. China, 2005, 17(5), 766
Chiou M. S., Chuang G. S., Chemosphere, 2006, 62(5), 731
Morais W. A., de Almeida A. L. P., Pereira M. R., Fonseca J. L. C., Carbohydr. Res., 2008, 343(14), 2489
Crini G., Gimbert F., Robert C., Martel B., Adam O., Morin-Crini N., De Giorgi F., Badot P. M., J. Hazard. Mater., 2008, 153(1), 96
Kittinaovarat S., Kansomwan P., Jiratumnukul N., Appl. Clay. Sci., 2010, 48(1), 87
Liu L., Wan Y. Z., Xie Y. D., Zhai R., Zhang B., Liu J., Chem. Eng. J., 2012, 187, 210
Zhang J. X., Zhou Q. X., Ou L. L., J. Chem. Eng. Data, 2011, 57(2), 412
Ji J. G., Zhang J. F., Hao S. L., Wu D. J., Liu L., Xu Y., Chem. Res. Chinese Universities, 2012, 28(1), 166
Zhang C. L., Hu X. M., Hou X. H., Tao Y. T., Chem. J. Chinese Universities, 2013, 34(3), 491
Bailey S. E., Olin T. J., Bricka R. M., Adrian D. D., Water Res., 1999, 33(11), 2469
Zhang Q. X., Liu H., Xu X. Q., Du M. X., Chem. J. Chinese Universities, 2012, 33(1), 107
O’Connor D. J., Connolly J. P., Water Res., 1980, 14(10), 1517
Voice T. C., Weber W. J., Environ. Sci. Technol., 1985, 19(9), 789
Di Toro D. M., Mahony J. D., Kirchgraber P. R., O’Byrne A. L., Pasquale L. R., Piccirilll D. C., Environ. Sci. Technol., 1986, 20(1), 55
Pan G., Liss P. S., J. Colloid Interface Sci., 1998, 201(1), 71
Helmy A. K., Ferreiro E. A., de Bussetti S. G., J. Colloid Interface Sci., 2000, 225(2), 398
Chang T. W., Wang M. K., Chemosphere, 2002, 48(4), 419
Wu X. F., Hu Y. L., Zhao F., Huang Z. Z., Lei D., J. Environ. Sci. China, 2006, 18(6), 1167
Fehse K. U., Borg H., Sorkau E., Pilchowski K., Luckner L., Water Air Soil Poll., 2010, 210(1/4), 211
Zhao L. X., Hou W. G., Colloids Surf. A, 2012, 396, 29
Zhao L. X., Song S. E., Du N., Hou W. G., Colloid Polym. Sci., 2013, 291(3), 541
Zhao L. X., Song S. E., Du N., Hou W. G., Acta Phys.-Chim. Sin., 2012, 28(12), 2905
Sorokin A. B., Quignard F., Valentin R., Mangematin S., Appl. Catal. A: Gen., 2006, 309(2), 162
Shen C. S., Song S. F., Zang L. L., Kang X. D., Wen Y. Z., Liu W. P., Fu L. S., J. Hazard. Mater., 2010, 177(1), 560
Ho Y. S., Carbon, 2004, 42(10), 2115
Ho Y. S., Pol. J. Environ. Stud., 2006, 15(1), 81
Zhang S. Q., Chin. J. Health Statistics, 2002, 19(1), 9
Iraolagoitia X. L. R., Martini M. F., Colloids Surf. B, 2010, 76(1), 215
Sheindorf C., Rebhun M., Sheintuch M., J. Colloid Interface Sci., 1981, 79(1), 136
Lu Y. F., Allen H. E., Environ. Pollut., 2006, 143(1), 60
Chiou M. S., Ho P. Y., Li H. Y., Dyes. Pigments, 2004, 60(1), 69
Huang R. H., Liu Q., Huo J., Yang B. C., Arab. J. Chem., 2013. doi: 10.1016/j.arabjc.201305017
Kim J. Y., Shin M. C., Park J. R., Nam K., J. Mater. Cycles Waste Manage., 2003, 5(1), 55
Hameed B. H., El-Khaiary M. I., J. Hazard. Mater., 2008, 154(1), 639
Xu C., Li W., Pan G., Acta Phys.-Chim. Sin., 2009, 25(9), 1737
Saha T. K., Karmaker S., Ichikawa H., Fukumori Y., J. Colloid Interface Sci., 2005, 286(2), 433
Zhao S. P., Zhou F., Li L. Y., Cao M. J., Zuo D. Y., Liu H. T., Compos. Part. B: Eng., 2012, 43(3), 1570
Uzun I., Güzel F., J. Colloid Interface Sci., 2004, 274(2), 398
Morais W. A., Fernandes A. L. P., Dantas T. N. C., Pereira M. R., Fonseca J. L. C., Colloid Surf. A, 2007, 310(1), 20
Chiou M. S., Li H. Y., Chemosphere, 2003, 50(8), 1095
Wang M. X., Wang Y. P., Tan W. F., Liu F., Feng X. H., Koopal L. K., J. Soil Sediment., 2010, 10(5), 879
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China(Nos.21173135, 51372141) and the Specialized Research Fund for the Doctoral Program of Higher Education of China(No.20110131130008).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Guo, Y., Hou, W., Liang, J. et al. Sorbent concentration effect on adsorption of methyl orange on chitosan beads in aqueous solutions. Chem. Res. Chin. Univ. 30, 837–843 (2014). https://doi.org/10.1007/s40242-014-4042-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-014-4042-x