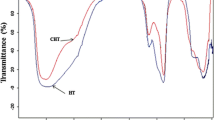
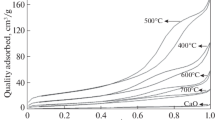
Abstract
An environmentally friendly heterogeneous catalyst, C60-doped TiO2(denoted as C60-TiO2), was prepared and firstly used to catalyze the synthesis of diphenyl carbonate(DPC) via the transesterification of phenol and dimethyl carbonate(DMC). The characterization results of FTIR, XRD, XPS, SEM, TEM and digital optical microscope(MIC) show that C60 promotes the dispersion of TiO2 and the prepared conditions affect the dispersion and the electron density of Ti species. The C60-TiO2-8[n(Ti):n(C60)=8:1] prepared with toluene and calcined at 200 °C exhibits the best catalytic performance. The DPC selectivity of 86.5% attained over C60-TiO2-8 was much higher than that(about 50%) attained over Ti-based catalysts reported, which was originated from the electronic effect of C60 and the efficient dispersion of Ti species by C60. The phenol conversion slowly decreased from 34.4% to 26.7% after four consecutive runs due to the leach of Ti species. It also suggested that the C60 could relieve the leach of Ti species compared with other Ti-based catalysts reported. Moreover, C60-TiO2-8 exhibited excellent catalytic behavior for disproportionation of methyl phenyl carbonate(MPC) with high DPC selectivity.
Similar content being viewed by others
References
Wang Z. Q., Yang X. G., Liu S. Y., Zhang H., Wang G. Y., Chem. Res. Chinese Universties, 2016, 32(3), 512
Haubrock J., Raspe M., Versteeg G. F., Kooijman H. A., Taylor R., Hogendoorn J. A., Ind. Eng. Chem. Res., 2016, 47, 9854
Deshmukh K. M., Qureshi Z. S., Dhake K. P., Bhanage B. M., Catal. Commun., 2010, 12, 207
Kim Y. T., Park E. D., Appl. Catal. A: Gen., 2009, 356, 211
Joshi U. A., Choi S. H., Jang J. S., Lee J. S., Catal. Lett., 2008, 123, 115
Luo S. W., Chen T., Tong D. S., Zeng Y., Lei Y. C., Wang G. Y., Chin. J. Catal., 2007, 28, 937
Zhou X., Ge X., Tang R. Z., Chen T., Wang G. Y., Chin. J. Catal., 2014, 35, 481
Cao M., Meng Y. Z., Lu Y. X., Catal. Commun., 2005, 6, 802
Li Z. H., Wang Y. J., Ding X. S., Zhao X. Q., Journal of Natural Gas Chemistry, 2009, 18, 104
Wang S. L., Tang R. Z., Zhang Y. Z., Chen T., Wang G. Y., Chem. Eng. Sci., 2015, 138, 93
Tong D. S., Yao J., Wang Y., Niu H. Y., Wang G. Y., J. Mol. Catal. A: Chem., 2007, 268, 120
Tang R. Z., Chen T., Chen Y., Zhang Y. Z., Wang G. Y., Chin. J. Catal., 2014, 35, 457
Kim W. B., Lee J. S., Catal. Lett., 1999, 59, 83
Wang S., Bai R., Mei F., Li G., Catal. Commun., 2009, 11, 202
Tong D. S., Chen T., Yao J., Wang Y., Wang G. Y., Shi D. C., Li Z., Chen Z. M., Chin. J. Catal., 2007, 28, 190
Meng Q. R., Bai J., Guo S. J., Li C. P., Chem. Res. Chinese Universi-ties, 2015, 31(6), 1072
Li Q. M., Chem. Res. Chinese Universities, 2013, 29(5), 1011
Tong H., Li H. L., Zhang X. G., Carbon, 2007, 45, 2424
Osuna S., Morera J., Cases M., Morokuma K., Sola M., J. Phys. Chem. A, 2009, 113, 9721
Niu F., Wu J., Zhang L., Li P., Zhu J., Wu Z., Song W., ACS Catal., 2011, 1, 1158
Nagashima H., Nakaoka A., Tajima S., Saito Y., Itoh K., Chem. Lett., 1992, 7, 1361
Beejapur H. A., Campisciano V., Franchi P., Lucarini M., Giacalone F., Chem. Cat. Chem., 2014, 6, 2419
Lin J., Zong R. L., Zhou M., Zhu Y. F., Appl. Catal. B: Environ., 2009, 89, 425
Yu J. G., Ma T. T., Liu G., Cheng B., Dalton Trans., 2011, 40, 6635
Zhang L., Wang Y., Xu T., Zhu S., Zhu Y., J. Mol. Catal. A: Chem., 2007, 331, 7
Yokosuka Y., Oki K., Nishikiori H., Tatsumi Y., Tanaka N., Fujii T., Res. Chem. Intermed., 2009, 35, 43
Bonelli B., Cozzolino M., Tesser R., Di Serio M., Piumetti M., Gar-rone E., Santacesari E., J. Catal., 2007, 246, 293
Ruoff R. S., Tse D. S., Malhotra R., Lorents D. C., J. Phys. Chem., 1993, 97, 3379
Li B., Tang R., Chen T., Wang G., Chin. J. Catal., 2012, 33, 603
Ge X., Li B. J., Hu J., Chen T., Wang G. Y., Hu X. T., Chem. Eng., 2011, 39, 061
Wang Q., Li C. H., Guo M., Luo S. J., Hu C. W., Inorg. Chem. Front., 2015, 2, 47
Coq B., Brotons V., Planeix J. M., de Menorval L. C., Dutartre R., J. catal., 1998, 176, 358
Luo Z., Wang Y., Zhang Y., Wei D., Jiang J., Wang G. Y., Fine Chem., 2008, 25, 813
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National High Technology Research and Development Program of China(No.2013AA031703).
Rights and permissions
About this article
Cite this article
Qu, Y., Yang, H., Wang, S. et al. High selectivity to diphenyl carbonate synthesized via transesterification between dimethyl carbonate and phenol with C60-doped TiO2 . Chem. Res. Chin. Univ. 33, 804–810 (2017). https://doi.org/10.1007/s40242-017-6500-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-017-6500-8