Abstract
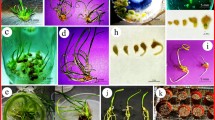
A rapid clonal propagation system for Kaempferia galanga (Zingiberaceae), a rare folk medicinal herb has been developed. Various concentrations of 6-benzyladenine (BA) and a range of auxins have been investigated for in vitro plantlet production, using rhizomes as explants. In vitro plantlet production has been achieved on 0.75 × Murashige and Skoog (MS) medium supplemented with 12 μM BA, 3 μM ∝-naphthaleneacetic acid (NAA) and 3% sucrose. The procedure ensures 13-fold rate of plantlet production every 4 weeks. Hardened plantlets produced normal storage roots as the parent plants. Around 1,000 plantlets have been produced successfully for field transfer.
Similar content being viewed by others
References
Avery GS, Burkholder PR & Creighton HB (1933) Production and distribution of growth hormone in shoots of Aesculus and Malus and its probable role in stimulating cambial activity. Amer J. Bot. 24: 51–58
Bonga JM & von Aderkas P (1992) In Vitro Culture of Trees. Kluwer Academic Publishers, Dordrecht
Bouillenne R & Went FW (1933) Recherches experimentales sur la neoformation des racines dans les plantules et les boutures des plantes superieures. Ann. Jard. Bot. Buiten. 43: 25–202
Cooke T (1905) The Flora of the Presidency of Bombay, Vol II. Taylor & Francis, London
Graf BA (1982) Exotica International, Pictorial Cyclopedia of Exotic Plants, Series 4, Vol 2. Roehrs Company Publishers, New Yersey
Hooker JD (1978) The Flora of British India, Vol VI. B/S Bishen Singh Mahendra Pal Singh, Dehradun, India
Kirtikar KR & Basu BD (1981) Indian Medicinal Plants, Vol IV. B/S Bishen Singh Mahendra Pal Singh, Dehradun, India
Murashige T & Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15: 473–497
Pence VC & Soukup VG (1993) Factors affecting the initiation of mini-rhizomes from Trillium erectum and T. grandiflorum tissues in vitro. Plant Cell Tiss. Org. Cult. 35: 229–235
Purohit SD, Dave A & Kukda G (1994) Micropropagation of safed musli (Chlorophytum borivilianum), a rare Indian medicinal herb. Plant Cell Tiss. Org. Cult. 39: 93–96
Romano A, Noronha C & Martins-Loucao MA (1995) Role of carbohydrates in micropropagation of cork oak. Plant Cell Tiss. Org. Cult 40: 159–167
Sato T, Kwon OC, Miyake H, Taniguchi T & Macda E (1995) Regeneration of plantlets from petiole callus of wild viola (Viola patrinii DC.). Plant Cell Rep. 14: 768–772
Sembdner G, Gross D, Liebisch HW & Schneider G (1980) Biosynthesis and metabolism of plant hormones. In: MacMillan J (ed) Encyclopedia of Plant Physiology (New Series), Vol 9 (pp 281–444). Springer-Verlag, Berlin
Sharma N, Chandel KPS & Paul A (1993) In vitro propagation of Gentiana kurroo – an indigenous threatened plant of medicinal importance. Plant Cell Tiss. Org. Cult. 34: 307–309
Vasil 1K (1991) Rationale for the scale up and automation of plant propagation. In: Vasil IK (ed) Scale up and Automation in Plant Propagation (pp 1–5). Academic Press Inc., London
Went FW (1929) On a substance causing root formation. Proc. Koninkl. Ned. Akad. Wetenschap. 32: 35–39
Went FW (1934) A test method for rhizocaline, the root forming substance. Proc. Koninkl. Ned. Akad. Wetenschap. 37: 445–455
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shirin, F., Kumar, S. & Mishra, Y. In vitro plantlet production system for Kaempferia galanga, a rare Indian medicinal herb. Plant Cell, Tissue and Organ Culture 63, 193–197 (2000). https://doi.org/10.1023/A:1010635920518
Issue Date:
DOI: https://doi.org/10.1023/A:1010635920518