Abstract
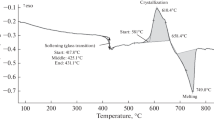
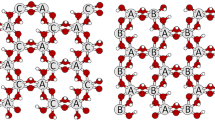
The degree of dehydroxylation of kaolinite, DTG and DIR, respectively, is characterized by thermogravimetric analysis (TG) and Fourier transform infrared spectroscopy (FTIR). The relation between DTG and DIR based on the infrared absorptions at 3600–3700, 915, 810, and 540 cm−1 is established. Three regions can clearly be distinguished: the dehydroxylation region (DTG<0.9), the metakaolinite region (0.9<DTG<1) and the ‘spinel’ region(DTG=1). The effect of the degree of dehydroxylation of kaolinite on the amount of reactive material is measured by the reaction enthalpy, ΔH, of the low-temperature reaction of the dehydroxylated kaolinite with a potassium silicate solution using differential scanning calorimetry (DSC). |ΔH| increases almost linearly with DTG in the dehydroxylation region. In the metakaolinite region, ΔH and thus the amount of reactive material, becomes constant. |ΔH| is sharply decreasing when metakaolinite transforms into other phases in the ‘spinel’ region. No significant differences in the reactivity of the dehydroxylates is detected with DSC. According to FTIR, the use of partially dehydroxylated kaolinite is not influencing the molecular structure of the low-temperature synthesized aluminosilicates, but residual kaolinite is retrieved as an additive.
Similar content being viewed by others
REFERENCES
R. H. Meinhold, R. C. T. Slade and T.W. Davies, Appl. Magn. Reson., 4 (1993) 141.
J. Rocha and J. Klinowski, Angew. Chem., 102 (1990) 539.
J. F. Lambert, W. S. Millman and J. J. Fripiat, J. Am. Chem. Soc., 111 (1989) 3517.
R. C. T. Slade, T. W. Davies, H. Atakul, R. M. Hooper and D. J. Jones, J. Mater. Sci., 27 (1992) 2490.
K. J. D. Mackenzie, I. W. M. Brown, R. H. Meinhold and M. E. Bowden, J. Amer. Ceram. Soc., 68 (1985) 293.
I. W. M. Brown, K. J. D. Mackenzie, M. E. Bowden and R. H. Meinhold, J. Amer. Ceram. Soc., 68 (1985) 298.
D. Massiot, P. Dion, J. F. Alcover and F. Bergaya, J. Amer. Ceram. Soc., 78 (1995) 2940.
R. L. Frost and A. M. Vassallo, Clays Clay Miner., 44 (1996) 635.
H. Rahier, B. Van Mele, M. Biesemans, J. Wastiels and X. Wu, J. Mater. Sci., 31 (1996) 71.
H. Rahier, B. Van Mele and J. Wastiels, J. Mater. Sci., 31 (1996) 80.
H. Rahier, W. Simons, B. Van Mele and M. Biesemans, J. Mater. Sci., 32 (1997) 2237.
H. Van Olphen and J. J. Fripiat, Data Handbook for Clay Materials and Other Non-Metallic Minerals, Pergamon Press, London 1979.
D. Potzold, B. Poppe and T. Trager, Silikattechnik, 38 (1985) 352.
A. Bachiorrini and M. Murat, C. R. Acad. Sci. Paris, 303 series II (20) (1986) 1783.
M. Murat, D. Mathurin, M. Driouche and A. Bachiorrini, Sci. Geol. Bull., 43 (1990) 213.
D. Mathurin, M.E. Chibi, and M. Murat, Thermochim. Acta, 98 (1986) 49.
P. R. Stuich, J. Amer. Ceram. Soc., 69 (1986) 61.
S. Swier, G. Van Assche, A. Van Hemelrijck, H. Rahier, E. Verdonck and B. Van Mele, J. Therm. Anal. Cal., 54 (1998) 585.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rahier, H., Wullaert, B. & Van Mele, B. Influence of the Degree of Dehydroxylation of Kaolinite on the Properties of Aluminosilicate Glasses. Journal of Thermal Analysis and Calorimetry 62, 417–427 (2000). https://doi.org/10.1023/A:1010138130395
Issue Date:
DOI: https://doi.org/10.1023/A:1010138130395