Abstract
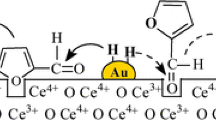
Based on the structure of characterization of Au/Fe2O3 catalysts, the mechanism of gold activation in supported gold catalysts was proposed as follow:
The possible catalytic active state is the partially oxidized gold (Auδ+) with unoccupied outer d orbitals, similar to the outer d orbital structure of Pt. Thus "inert" gold can become very active for CO oxidation.
Similar content being viewed by others
REFERENCES
J. Schwank: Gold Bull., 16, 103 (1983).
M. Haruta, N. Yamada, T. Kobayashi, S. Lijima: J. Catal., 115, 301 (1989).
S.D. Gardner, G.B. Hoflund, B.T. Upchurch, D.R. Schryer, E.J. Kielin, J. Schryer: J. Catal., 129, 114 (1991).
S.K. Tanielyan, R.L. Augustine: Appl. Catal. A., 85, 73 (1992).
A. Knell, P. Barnickel, A. Baiker, A. Wokaun: J. Catal., 137, 306 (1992).
Naomi Funazki, Akihide Hemmi: Sensors Actuators B., 13, 536 (1993).
Zhengping Hao, Lidun An, Hongli Wang: Chin. Chem. Lett., 5, 447 (1995).
M. Haruta: Now & Future., 7, 13 (1992).
Lidun An, Zhengping Hao: China Pat.,CN 1125638.
Zhengping Hao, Lidun An Junling Zhou, Hongli Wang: React. Kinet. Catal. Lett., 59, 259 (1995).
Zhengping Hao, Lidun An, Hongli Wang: J. MoL Catal. (Chinese), 9, 233 (1995).
Zhengping Hao, Lidun An, Hongli Wang: J. Mol. Catal. (Chinese), 11 51 (1997).
Hongwei Xiang, Tiandou Hu: J. Chem. Phys. (Chinese), 18, 75 (1995).
Caixia Qi, Tingfang Bai, Lidun An: React. Kinet. Catal. Lett., 54, 131 (1995).
C.D. Wagner, W.M. Riggs, LE.Davis, J.T. Moulder, G.E. Muilenberg: Eds, Handbook of X-ray Photoelectron Spectroscopy, p. 154. Perkin-Elmer Corporation 1979.
S. Imamura, H. Sawada, K. Vemura: J. Catal., 109, 198 (1988).
Qun Tong:. J. Inst Daging. Petroleum (Chinese) 16, 60 (1992).
Rights and permissions
About this article
Cite this article
Hao, Z., An, L., Wang, H. et al. Mechanism of Gold Activation in Supported Gold Catalysts for CO Oxidation. Reaction Kinetics and Catalysis Letters 70, 153–160 (2000). https://doi.org/10.1023/A:1010383220836
Issue Date:
DOI: https://doi.org/10.1023/A:1010383220836