Abstract
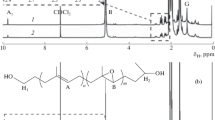
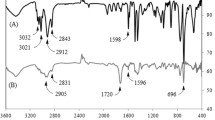
Four polyhedral oligomeric silsesquioxane (POSS) macromers, R7Si8O12styrene (R=isobutyl, cyclopentyl, cyclohexyl, or phenyl), containing a single polymerizable styrene unit were synthesized from the POSS-trisilanols R7Si7O9(OH)3 in a high yield and purity. The base-assisted reaction to produce these macromers appears to be general for POSS-trisilanols of this type. However, in some cases it may be necessary to control the rate at which the base is introduced during reaction to prevent unwanted side reactions that decompose the trisilanol. Cyclohexyl-, cyclopentyl-, and isobutyl-substituted POSS-stryenes (at 30 wt% or approximately 4 mol% loadings) undergo free radical bulk polymerizations with styrene to produce polymers that show variation in the modulus above the glass transition temperature. The phenylPOSS derivative is too insoluble in styrene to undergo this polymerization. The effect on the modulus is more pronounced for the copolymers containing 30 wt% cycloalkylPOSS than that seen with isobutylPOSS, which is similar to that for bulk polymerized polystyrene. The effect of the group at the POSS cage on bulk polymer properties has been noted before and is presumed to arise from differences in polymer microstructure.
Similar content being viewed by others
REFERENCES
A recent issue of Chemistry of Materials is devoted entirely to nanocomposites and includes 35 reviews on various aspects of this field. Chem. Mater. 13, 3059 (2001).
C. Sanchez, G. J. de A. A. Soler-Illia, F. Ribot, T. Lalot, C. R. Mayer, and V. Cabuil, Chem. Mater. 13, 3061 (2001). (b) R. M. Laine, C. Sanchez, C. J. Brinker, and E. Giannelis (eds.), Organic/Inorganic Hybrid Materials, MRS Symposium Proceedings (1998), Vol. 519. (c) L. C. Klein, L. F. Francis, M. R. De Guire, and J. E. Mark (eds.), Organic/Inorganic Hybrid Materials II, MRS Symposium Proceedings (1999), Vol. 576. (d) R. M. Laine, C. Sanchez, C. J. Brinker, and E. Giannelis (eds.), Organic/Inorganic Hybrid Materials-2000, MRS Symposium Proceedings (2000), Vol. 628.
J. J. Schwab and J. D. Lichtenhan, Appl. Organomet. Chem. 12, 707 (1998). J. D. Lichtenhan, Comments Inorg. Chem. 17, 115 (1995).
A. Romo-Uribe, P. T. Mather, T. S. Haddad, and J. D. Lichtenhan, J. Polym. Sci. Polym. Phys. 36, 1857 (1998). (b) T. S. Haddad and J. D. Lichtenhan, Macromolecules 29, 7302 (1996).
A. B. Pangborn, M. A. Giardello, R. H. Grubbs, R. K. Rosen, and F. J. Timmers, Organometallics 15, 1518 (1996).
F. J. Feher, R. Terroba, and J. W. Ziller, Chem. Commun. 2309 (1999). (b) V. Ruffieux, G. Schmid, P. Braunstein, and J. Rose, Chem. Eur. J. 3, 900 (1997). (c) F. J. Feher, D. A. Newman, and J. F. Walzer, J. Am. Chem. Soc. 111, 1741 (1989). (d) F. J. Feher, T. A. Budzichowski, R. L. Blanski, K. L. Weller, and J. W. Ziller, Organometallics 10, 2526 (1991). (e) J. F. Brown Jr. and L. H. Vogt, Jr., J. Am. Chem. Soc. 87, 4313 (1965).
Hybrid Plastics LLC: www.hybridplastics.com
S. Rubinsztajn and J. Stein, J. Inorg. Organomet. Polym. 5, 43 (1995).
F. J. Feher and D. A. Newman, J. Am. Chem. Soc. 112, 1931 (1990).
E. G. Shockey, A. G. Bolf, P. F. Jones, J. J. Schwab, K. P. Chaffee, T. S. Haddad, and J. D. Lichtenhan, Appl. Organometal. Chem. 13, 1 (1999).
Other groups that have been sucessfully incorporated include norbornenyl and acrylic functionalities.
The pKa of the cyclopentylPOSS trisilanol is reported to be 7.6. R. Duchateau, U. Cremer, R. J. Harmsen, S. I. Mohamud, H. C. L. Abbenhuis, R. A. van Santen, A. Meetsma, S. K.-H. Thiele, M. F. H. van Tol, and M. Kranenburg, Organometallics 18, 5447 (1999).
F. J. Feher, J. J. Schwab, D. Soulivong, and J. W. Ziller, Main Group Chem. 2, 123 (1997).
T. S. Haddad, B. M. Moore, and S. H. Phillips, Polym. Prepr. 42(1), 196 (2001).
L. Zheng, R. J. Farris, and E. B. Coughlin, Macromolecules 34, 8034 (2001). (b) B. X. Fu, B. S. Hsiao, S. Pagola, P. Stephens, H. White, M. Rafailovich, J. Sokolov, P. T. Mather, H. G. Jeon, S. Phillips, J. Lichtenhan, and J. Schwab, Polymer 42, 599 (2001). (c) H. G. Jeon, P. T. Mather, and T. S. Haddad, Polym. Int. 49, 453 (2000). (d) P. T. Mather, H. G. Jeon, A. Romo-Uribe, T. S. Haddad, and J. D. Lichtenhan, Macromolecules 32, 1194 (1999). (e) A. Lee and J. D. Lichtenhan, Macromolecules 31, 4970 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haddad, T.S., Viers, B.D. & Phillips, S.H. Polyhedral Oligomeric Silsesquioxane (POSS)-Styrene Macromers. Journal of Inorganic and Organometallic Polymers 11, 155–164 (2001). https://doi.org/10.1023/A:1015237627340
Issue Date:
DOI: https://doi.org/10.1023/A:1015237627340