Abstract
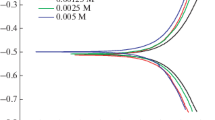
The corrosion of cadmium in 0.5m HClO4, CH3COOH or 0.25m H2SO4 in the absence and presence of small amounts of phenothiazine (10−6–10−4 m) as an inhibitor has been studied using both electrochemical impedance spectroscopy (EIS) and Tafel-plot techniques. Measurements were carried out at cathodic, open circuit and anodic potentials at different temperatures. In HClO4 and H2SO4 solutions the inhibition efficiency increases as both the concentration of phenothiazine and the temperature are increased. The effect of temperature in CH3COOH solution gives an opposite effect, that is, a decrease in the inhibition efficiency with increasing temperature. This behaviour is attributed to competitive adsorption between the inhibitor molecules and the acetate ions at the metal–solution interface. Phenothiazine can be considered as an inhibitor of mixed type with more pronounced effects in the anodic direction. The thermodynamic parameters in HClO4 containing the inhibitor were evaluated using the Bockris–Swinkels adsorption isotherm with n=4 as the configurational function. These parameters reveal that phenothiazine is adsorbed on the cadmium surface in molecular form with a strong interaction between the metal and the inhibitor.
Similar content being viewed by others
References
S. Tamil Selvan, R. Sabapathi and N. Venkatakrishnan, J. Appl. Electrochem. 21 (1991) 646.
M. S. Abdel Aal, A. A. Abdel-Wahab and F. H. Assaf, Metalloberflache 34 (1980) 323.
M. S. Abdel Aal and F. H. Assaf, Trans. SAEST 15 (1980) 107.
M. S. Abdel Aal, S. Radwan and A. El-Sayed, Br. Corros. J. 18 (1983) 102.
S. S. Abdel Rehim, A. El-Sayed and A. A. Samahi, Surf. Coat. Technol. 27 (1986) 205.
F. Rashwan, A. El-Sayed and H. Mohran, Bull. Electrochem. India 9 (1993) 57.
K. Aramaki, M. Hagiwara and H. Nishihara, Corros. Sci. 27 (1987) 487.
I. Sekine, M. Sanbongi, H. Hagiuda, T. Oshibe, M. Yuasa, T. Imahama, Y. Shibata and T. Wake, J. Electrochem. Soc. 139 (1992) 3167.
S. L. Granese and B. M. Rosales, in Proceedings of 7th European Symposium on Corrosion Inhibitors, Ferrara, Italy, vol. 1 (1990) p. 73.
T. P. Hoar and R. D. Holliday, J. Appl. Chem. 3 (1953) 502.
S. Sankarapapavinasam and M. F. Ahmed, J. Appl. Electrochem. 22 (1992) 390.
G. Mrowczynski and Z. Szklarska Smialowska, ibid. 9 (1979) 201.
L. I. Antropov, 'Theoretical Electrochemistry', Mir Publications, Moscow (1972) p.276.
L. I. Antropov, Corros. Sci. 7 (1967) 607.
H. B. Rudresh and S. M. Mayanna, J. Electrochem. Soc. 124 (1977) 340.
J. O'M. Bockris, M. A. V. Devanathan and K. Muller, Proc. R. Soc., London A274 (1963) 55.
J. O'M. Bockris and D. A. J. Swinkels, J. Electrochem. Soc. 111 (1964) 736.
B. G. Ateya, J. Electroanal. Chem. 76 (1977) 191.
R. Gurney, `Ionic Processes in Solution' Dover, New York (1953) p. 48.
D. B. Damaskin, Electrochimia 3 (1976) 1390.
R. K. Dinnappa and S. M. Mayanna, Corrosion 38 (1982) 525.
M. J. Lampinen and M. Fomino, J. Electrochem. Soc. 140 (1993) 3537.
R. K. Dinnappa and S. M. Mayanna, J. Appl. Electrochem. 11 (1981) 111.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
EL-SAYED , A. Phenothiazine as inhibitor of the corrosion of cadmium in acidic solutions. Journal of Applied Electrochemistry 27, 193–200 (1997). https://doi.org/10.1023/A:1018456008267
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1018456008267