Abstract
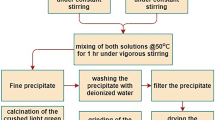
Fine particles of willemite, α-Zn2SiO4, were prepared by both solution combustion and sol-gel methods. Both processes yield single-phase, large-surface area (26- and 78-m2/g), sinteractive willemite powders. Thermal evolution of crystalline phases was studied using X-ray powder diffraction patterns. The combustion method favors low-temperature formation of willemite compared to the sol-gel method. The powders, when uniaxially pressed and sintered at 1300°C, achieved 78–80% theoretical density. The microstructures of the sintered body show the presence of equiaxed 0.5- to 4-μm grains. Blue pigments of willemite doped with Co2+ and Ni2+ were also prepared by the combustion process.
Similar content being viewed by others

REFERENCES
J. A. Speer and P. H. Ribbe, in Reviews in Mineralogy, Vol. 5, 2nd ed., P. H. Ribbe, ed. (Mineralogical Society of America, Washington, DC, 1982), p. 429.
K. C. Mishra, K. H. Jhonson, B. G. Deboer, J. K. Berkowitz, J. Olsen, and E. A. Dale, Luminescence 47, 197–206 (1991).
C. Bartohu, J. Benoit, P. Benalloul, and A. Morell, J. Electrochem. Soc. 141(2), 524–528 (1994).
Z. Strand, Glass Science and Technology, Vol. 8 (Elsevier, Amsterdam, 1986), p. 101.
D. Lepkova, A. Nenor, I. Pavlova, et al., Stroit. Mater. Silik. Prom. st. 21(2), 18–20 (1980).
J. Alarcon, P. Escribano, and R. Ma. Marin, Br. Ceram. Trans. J. 84, 170–172 (1985).
E. Rosello, A. Borrio, M. Guillem, and M. Cormen, Br. Ceram. Trans. J. 84, 175–177 (1985).
C. C. Lee, P. Shen, and H. Y. Lu, J. Mater. Sci. 24(9), 3300–3304 (1989).
A. Majumdar and D. Ganguli, Trans. Ind. Ceram. Soc. 50(4), 93–97 (1991).
J. J. Kingsley and K. C. Patil, Mater. Lett. 6, 427–431 (1988).
R. G. Chandran, K. C. Patil, and G. T. Chandrappa, Int. J. Self.-Prop. High-Temp. Synth. 3, 131–142 (1994).
R. G. Chandran, K. C. Patil, and G. T. Chandrappa, J. Mater. Sci. Lett. 14, 548–551 (1995).
G. T. Chandrappa, R. G. Chandran, and K. C. Patil, Int. J. Self-Prop. High-Temp. Synth. 4, 183–191 (1995).
C. Ainsworth and R. Jones, J. Am. Chem. Soc. 77(3), 621–624 (1955).
S. R. Jain, K. C. Adiga, and V. R. Paiverneker, Combust. Flame 40, 71–76 (1981).
Ya. Ya. Kharitonov, R. I. Machkhoshvili, and L. V. Goeva, Koord. Khim. 5, 985–994 (1979).
M. S. Hegde, S. S. Monoharan, and K. C. Patil, in Proc. Eighth Natl. Symp. Therm. Anal., Bhubaneshwar, India (1991), pp. 578–580.
C. C. Lin and P. Shen, J. Solid State Chem. 112, 381–386 (1994).
R. W. G. Wyckoff, Cryst. Struct. 3, 134 (1963).
R. A. Nyquist and R. O. Kagel, Infrared Spectra of Inorganic Compounds (Academic Press, New York, 1971).
G. Engelhardt and D. Michel, High-Resolution Solid-State NMR of Silicates and Zeolites (John Wiley and Sons, Chichester, 1987), p. 159.
T. Bates, Ligand Field Theory and Absorption Spectra of Transition Metals Ions Glass. Modern Aspects of Vitreous State—2 (Mackenzie, Butterworths, London, 1962).
T. C. Brunold, H. U. Gudel, and E. Cavalli, Chem. Phys. Lett. 268, 413–420 (1997).
T. C. Brunold, H. U. Gudel, and E. Cavalli, Chem. Phys. Lett. 252, 112–120 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chandrappa, G.T., Ghosh, S. & Patil, K.C. Synthesis and Properties of Willemite, Zn2SiO4, and M2+:Zn2SiO4 (M = Co and Ni). Journal of Materials Synthesis and Processing 7, 273–279 (1999). https://doi.org/10.1023/A:1021816803246
Issue Date:
DOI: https://doi.org/10.1023/A:1021816803246