Abstract
Purpose. We assessed the application of water-soluble polymer-based nanofibers prepared by electrostatic spinning as a means of altering the dissolution rate of the poorly water-soluble drug, itraconazole.
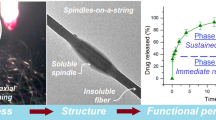
Methods. Organic solvent-based solutions of itraconazole/HPMC mixtures were electrostatically spun at 16 and 24 kV. The formed nanofibers were collected as a non-woven fabric. The samples were analyzed by scanning electron microscopy, differential scanning calorimetry, and dissolution rate.
Results. Scanning electron microscopy showed fiber diameters of 1-4 μm and 300-500 nm depending on the applied voltage. Differential scanning calorimetry measurements found that the melting endotherm for itraconazole was not present, suggesting the formation of an amorphous solid dispersion or solution. Dissolution studies assessed several presentations including direct addition of the non-woven fabrics to the dissolution vessels, folding weighed samples of the materials into hard gelatin capsules and placing folded material into a sinker. Controls included a physical mixture as well as solvent cast and melt extruded samples. Electrospun samples dissolved completely over time with the rate of dissolution depending on the formulation presentation and drug to polymer ratio. The physical mixture did not appreciably dissolve in these conditions.
Conclusions. The application of electrostatic spinning to pharmaceutical applications resulted in dosage forms with useful and controllable dissolution properties.
Similar content being viewed by others
REFERENCES
R. A. Prentis, Y. Lis, and S. R. Walker. Pharmaceutical innovation by seven UK-owned pharmaceutical companies (1964-1985). Br. J. Clin. Pharmacol. 25:387-396 (1988).
C. A. Lipinski, F. Lombardo, B. W. Dominy, and P. J. Feeney. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 23:3-25 (1997).
C. A. Lipinski. Avoiding investment in doomed drugs. Curr. Drug Discov. 1:17-19 (2001).
A. A. Noyes and W. R. Whitney. The rate of solution of solid substances in their own solutions. J. Am. Chem. Soc. 19:930-934 (1897).
D. E. Wurster and P. W. Taylor. Dissolution rates. J. Pharm. Sci. 54:169-175 (1965).
S. M. Grant and S. P. Clissold. Itraconazole. Drugs 37:310-344 (1989).
K. De Beule. and J. Van Gestel. Pharmacology of itraconazole. Drugs 61(Suppl. 1):27-33 (2001).
S. Jain and V. Seghal. Itraconazole: an effective oral antifungal for onchomychosis. Int. J. Dermatol. 40:1-5 (2001).
J. Peeters, P. Neeskens, J. P. Tollenaere, P. Van Remoortere, and M. E. Brewster. Characterization of the interaction of 2-hydroxypropyl-β-cyclodextrin with itraconazole at pH 2, 4 and 7. J. Pharm. Sci. 91:1414-1422 (2002).
P. Sheen, V. Khetarpal, C. Cariola, and C. Rowlings. Formulation of a poorly water-soluble drug in solid dispersions to improve bioavailability. Int. J. Pharm. 118:221-227 (1995).
A. T. Serajuddin. Solid dispersion of poorly water-soluble drugs. Early promises, subsequent problems and recent breakthroughs. J. Pharm. Sci. 88:1058-1066 (1999).
C. Leuner. and J. Dressman. Improving drug solubility for oral delivery using solid dispersion. Eur. J. Pharm. Biopharm. 50:47-60 (2000).
D. Erkoboni and R. Andersen. Improved aqueous solubility pharmaceutical formulation. World Patent 0056726 (2000).
P. A. Gilis, V. De Conde, and R. Vandecruys. Beads having a core coated with an antifungal and a polymer. US Patent 5633015 (1997).
L. Baert, D. Thone, and G. Verreck. Antifungal compositions with improved bioavailability. World Patent 9744014 (1997).
D. H. Reneker and I. Chun. Nanometre diameter of polymer, produced by electrospinning. Nanotechnology 7:216-223 (1996).
J. Doshi and D. H. Reneker. Electrospinning process and applications of electrospun fibers. J. Electrostatics 35:151-160 (1995).
E. R. Kenawy, G. L. Bowlin, K. Mansfield, J. Layman, D. G. Simpson, E. H. Sanders, and G. E. Wnek. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Control. Release 81:57-64 (2002).
F. Ignatious and J.M. Baldoni. Electrospun pharmaceutical compositions. World Patent 0154667 (2001).
A. L. Yarin, S. Koombhongse, and D. H. Reneker. Taylor cone and jetting from liquid droplets in electrospinning of nanofibers. J. Appl. Phys. 90:4836-4846 (2001).
A. L. Yarin, S. Koombhongse, and D. H. Reneker. Bending instability in electrospinning of nanofibers. J. Appl. Phys. 89:3018-3026 (2001).
J. M. Deitzel, J. Kleinmeyer, D. Harris, and N. C. Beck Tan. The effect of processing variables on the morphology of electrospun nanofibers and textiles. Polymers 42:261(2001).
D. S. Kath, K. W. Robinson, M. A. Attawia, F. K. Ko, and C. T. Laurencin. Bioresorbable nanofiber based systems for wound healing: optimization of fabrication parameters. Transactions of the 28th Annual Meeting for the Society for Biomaterials, 143(2002).
K. Six, G. Verreck, J. Peeters, K. Binnemans, K. Bergmans, P. Augustijns, R. Kinget, and G. Van den Mooter. Investigation of thermal properties of glassy itraconazole: identification of monotropic mesophase. Thermochim. Acta 376:175-181 (2001).
F. N. Kelley and F. Bueche. Viscosity and glass-transition temperature relations for polymer-dilute systems. J. Poly. Sci. 50:549-556 (1961).
M. Gordon and J. S. Taylor. Ideal copolymers and the second-order transitions of synthetic rubbers. I. Noncrystalline copolymers. J. Appl. Chem. 2:493-500 (1952).
R. Simha and R. F. Boyer. General relation involving the glass temperature and coefficients of expansion of polymers. J. Chem. Phys. 37:1003-1007 (1962).
T. Kissel, M. A. Rummelt, and H. P. Bier. Wirkstoffreisetzung aus bioabbaubaren Mikropartikeln. Dtsch. Apoth. Ztg. 133:29-32 (1993).
G. Verreck, L. Baert, J. Peeters, and M. Brewster. Improving aqueous solubility and bioavailability for itraconazole by solid dispersion approach. AAPSPharmSci 3:M2157(2001).
K. Six, C. Leuner, J. Dressman, G. Verreck, J. Peeters, N. Blaton, P. Augustijns, R. Kinget, and G. Van den Mooter. Thermal properties of hot-stage extrudates of itraconazole and Eudragit E100. Phase separation and polymorphism. J. Thermal Anal. Calorimetry 68:591-601 (2002).
L. Baert, J. Peeters and G. Verreck. Solid mixtures of cyclodextrins prepared via melt-extrusion. World Patent 9718839 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Verreck, G., Chun, I., Peeters, J. et al. Preparation and Characterization of Nanofibers Containing Amorphous Drug Dispersions Generated by Electrostatic Spinning. Pharm Res 20, 810–817 (2003). https://doi.org/10.1023/A:1023450006281
Issue Date:
DOI: https://doi.org/10.1023/A:1023450006281