Abstract
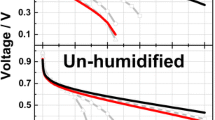
The current dependence of the ohmic resistance of Nafion membranes was examined with different types of humidification: cathodic (ChAd), anodic (CdAh), anodic and cathodic (ChAh) and no humidification at all (CdAd). Data show that for stacks with humidified cathodes (ChAd and ChAh), the resistance is small and relatively insensitive to the presence of the anodic humidification. On the contrary, for stacks with non-humidified cathodes (CdAh and CdAd), the membrane resistance is high and strongly dependent on current and anodic humidification. The kinetics of membrane dehydration was examined by recording the galvanostatic transients of the stack voltage and resistance, after removing the humidification. It was found that the changes in the ohmic resistance ΔR Ω(t), although significant, cannot explain entirely the observed decay of the stack voltage. To account for the difference, an additional resistive term is introduced ΔR p(t). Explicit equations were found for the time and current dependence of the two resistive terms ΔR Ω(t) and ΔR p(t) after humidification removal. A tentative explanation for the new resistive term was provided using electrochemical impedance spectroscopy (EIS). EIS data obtained at low overpotential show that dehydration of the Nafion present in the cathode catalytic layer results in an increase of the polarization resistance; the apparent deactivation of the cathode electrocatalyst appears to be due to a decrease of the electrochemically active surface area.
Similar content being viewed by others
References
T.A. Zawodzinski, C. Derouin, S. Radzinski, R.J. Sherman, V.T. Smith, T.E. Springer and S. Gottesfeld, J. Electrochem. Soc. 140 (1993) 1041.
J.T. Hinatsu, M. Mizuhata and H. Takenada, J. Electrochem. Soc. 141 (1994) 1493.
T.A. Zawodzinski, T.E. Springer, J. Davey, R. Jestel, C. Lopez, J. Valerio and S. Gottesfeld, J. Electrochem. Soc. 140 (1993) 1981.
M.W. Verbrugge and R.F. Hill, J. Electrochem. Soc. 137 (1990) 3770.
V.A. Paganin, C.L.F. Oliveira, E.A. Ticianelli, T.E. Springer and E.R. Gonzalez, Electrochim. Acta 43 (1998) 3761.
T. Okada, G. Xie and Y. Tanabe, J. Electroanal. Chem. 413 (1996) 49.
T.E. Springer, T.A. Zawodzinski and S. Gottesfeld, J. Electrochem. Soc. 138 (1991) 2334.
K. Dannenberg, P. Ekdunge and G. Lindbergh, J. Appl. Electrochem. 30 (2000) 1377.
D.R. Sena, E.A. Ticianelli, V.A. Paganin and E.R. Gonzalez, J. Electroanal. Chem. 477 (1999) 164.
G.J.M. Jansen and M.L.J. Overvelde, J. Power Sources 101 (2001) 117; R.J. Bellows, M.Y. Lin, M. Arif, A.K. Thompson and D. Jacobson, J. Electrochem. Soc. 146 (1999) 1099.
T.A. Zawodizinski, M. Neeman, L.O. Sillerud and S. Gottesfeld, J. Phys. Chem. 95 (1991) 1040.
P.C. Reike and N.E. Vanderborgh, J. Membr. Sci. 32 (1987) 313.
S. Slade, S.A. Campbell, T.R. Ralph and F.C. Walsh, J. Electrochem. Soc. 149 (2002) A 1556.
F.N. Buchi and G.G. Scherer, J. Electroanal. Chem. 404 (1996) 37.
F.N. Buchi and G.G. Scherer, J. Electrochem. Soc. 148 (2001) A 183.
A. Paganin, E.A. Ticianelli and E.R. Gonzalez, in S. Gottesfeld, G. Halpert and A. Landgrebe (Eds) ‘Proton Conducting Membrane Fuel Cells’, PV 95-23, The Electrochem. Soc. Proceedings Series (Pennington, NJ, 1995), p. 102.
M. Watanabe, H. Igarashi, H. Uchida and F. Hirashawa, J. Electroanal. Chem. 399 (1995) 239.
M. Ciureanu and M. Badita, J. New Mat. Electrochem. Syst. in press.
M. Ciureanu and R. Roberge, J. Phys. Chem. 105, (2001) 3531.
C. Hawk, A. Smirnova, J.M. Fenton and H.R. Kunk, Proceedings of the 200th Meeting of the Electrochemical Society, San Francisco, 2–7 Sept. (2001), Abstr. 424.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ciureanu, M. Effects of Nafion® Dehydration in PEM Fuel Cells. Journal of Applied Electrochemistry 34, 705–714 (2004). https://doi.org/10.1023/B:JACH.0000031102.32521.c6
Issue Date:
DOI: https://doi.org/10.1023/B:JACH.0000031102.32521.c6