Abstract
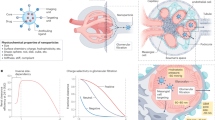
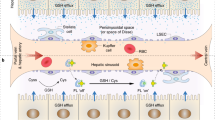
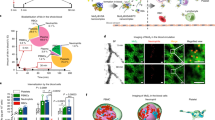
The field of nanotechnology holds great promise for the diagnosis and treatment of human disease. However, the size and charge of most nanoparticles preclude their efficient clearance from the body as intact nanoparticles. Without such clearance or their biodegradation into biologically benign components, toxicity is potentially amplified and radiological imaging is hindered. Using intravenously administered quantum dots in rodents as a model system, we have precisely defined the requirements for renal filtration and urinary excretion of inorganic, metal-containing nanoparticles. Zwitterionic or neutral organic coatings prevented adsorption of serum proteins, which otherwise increased hydrodynamic diameter by >15 nm and prevented renal excretion. A final hydrodynamic diameter <5.5 nm resulted in rapid and efficient urinary excretion and elimination of quantum dots from the body. This study provides a foundation for the design and development of biologically targeted nanoparticles for biomedical applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hardman, R. A toxicologic review of quantum dots: toxicity depends on physicochemical and environmental factors. Environ. Health Perspect. 114, 165–172 (2006).
Ballou, B. et al. Sentinel lymph node imaging using quantum dots in mouse tumor models. Bioconjug. Chem. 18, 389–396 (2007).
Uyeda, H.T., Medintz, I.L., Jaiswal, J.K., Simon, S.M. & Mattoussi, H. Synthesis of compact multidentate ligands to prepare stable hydrophilic quantum dot fluorophores. J. Am. Chem. Soc. 127, 3870–3878 (2005).
Chapman, A.P. et al. Therapeutic antibody fragments with prolonged in vivo half-lives. Nat. Biotechnol. 17, 780–783 (1999).
Goel, A. et al. Genetically engineered tetravalent single-chain Fv of the pancarcinoma monoclonal antibody CC49: improved biodistribution and potential for therapeutic application. Cancer Res. 60, 6964–6971 (2000).
Fu, A. et al. Semiconductor quantum rods as single molecule fluorescent biological labels. Nano Lett. 7, 179–182 (2007).
Mammen, M., Choi, S.K. & Whitesides, G.M. Polyvalent interactions in biological systems: implications for design and use of multivalent ligands and inhibitors. Angew. Chem. Int. Ed. Engl. 37, 2754–2794 (1998).
Peng, Z.A. & Peng, X. Formation of high-quality CdTe, CdSe, and CdS nanocrystals using CdO as precursor. J. Am. Chem. Soc. 123, 183–184 (2001).
Dabbousi, B.O. et al. (CdSe)ZnS core-shell quantum dots: synthesis and characterization of a size series of highly luminescent nanocrystallites. J. Phys. Chem. B 101, 9463–9475 (1997).
Fisher, B.R., Eisler, H.-J., Stott, N.E. & Bawendi, M.G. Emission intensity dependence and single-exponential behavior in single colloidal quantum dot fluorescence lifetimes. J. Phys. Chem. B 108, 143–148 (2004).
Mattoussi, H. et al. Self-assembly of CdSe-ZnS quantum dot bioconjugates using an engineered recombinant protein. J. Am. Chem. Soc. 122, 12142–12150 (2000).
Frangioni, J.V., Kim, S.W., Ohnishi, S., Kim, S. & Bawendi, M.G. Sentinel lymph node mapping with type-II quantum dots. Methods Mol. Biol. 374, 147–160 (2007).
Misra, P., Humblet, V., Pannier, N., Maison, W. & Frangioni, J.V. Production of multimeric prostate-specific membrane antigen small molecule radiotracers using a solid-phase 99mTc pre-loading strategy. J. Nucl. Med. 48, 1379–1389 (2007).
Humblet, V., Misra, P. & Frangioni, J.V. An HPLC/mass spectrometry platform for the development of multimodality contrast agents and targeted therapeutics: prostate-specific membrane antigen small molecule derivatives. Contrast Media Mol. Imaging 1, 196–211 (2006).
Kim, S. et al. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nat. Biotechnol. 22, 93–97 (2004).
De Grand, A.M. & Frangioni, J.V. An operational near-infrared fluorescence imaging system prototype for large animal surgery. Technol. Cancer Res. Treat. 2, 553–562 (2003).
Nakayama, A., del Monte, F., Hajjar, R.J. & Frangioni, J.V. Functional near-infrared fluorescence imaging for cardiac surgery and targeted gene therapy. Mol. Imaging 1, 365–377 (2002).
Pappenheimer, J.R., Renkin, E.M. & Borrero, L.M. Filtration, diffusion and molecular sieving through peripheral capillary membranes; a contribution to the pore theory of capillary permeability. Am. J. Physiol. 167, 13–46 (1951).
Prescott, L.F., McAuslane, J.A. & Freestone, S. The concentration-dependent disposition and kinetics of inulin. Eur. J. Clin. Pharmacol. 40, 619–624 (1991).
Olmsted, S.S. et al. Diffusion of macromolecules and virus-like particles in human cervical mucus. Biophys. J. 81, 1930–1937 (2001).
Hansen, N.E., Karle, H. & Andersen, V. Lysozyme turnover in the rat. J. Clin. Invest. 50, 1473–1477 (1971).
Lund, U. et al. Glomerular filtration rate dependence of sieving of albumin and some neutral proteins in rat kidneys. Am. J. Physiol. Renal Physiol. 284, F1226–F1234 (2003).
Solomon, A., Waldmann, T.A., Fahey, J.L. & McFarlane, A.S. Metabolism of Bence Jones Proteins. J. Clin. Invest. 43, 103–117 (1964).
Bradwell, A.R., Carr-Smith, H.D., Mead, G.P., Harvey, T.C. & Drayson, M.T. Serum test for assessment of patients with Bence Jones myeloma. Lancet 361, 489–491 (2003).
Acknowledgements
The Biophysical Instrumentation Facility for the Study of Complex Macromolecular Systems (NSF-0070319 and NIH GM68762) is gratefully acknowledged. This work was supported in part by the US National Science Foundation–Materials Research Science and Engineering Center Program under grant DMR-9808941 (M.G.B.), National Institutes of Health (NIH) grant no. R21/R33 EB-000673 (J.V.F. and M.G.B.), and a fellowship from the Charles A. King Trust, Bank of America, Co-Trustee (H.S.C.). M.G.B. also acknowledges support from the NIH-funded Massachusetts Institute of Technology–Harvard NanoMedical Consortium (1U54-CA119349, a Center of Cancer Nanotechnology Excellence). We thank Barbara L. Clough for medical editing and Grisel Vazquez for administrative assistance.
Author information
Authors and Affiliations
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6; Supplementary Methods; Supplementary Video 1 (PDF 1020 kb)
Supplementary Video 1
Real-time excretion of intravenously injected QD515 (10 pmol/g body weight) into the urinary system of the rat over 9 seconds, starting at 1 hour post-injection. (MOV 1632 kb)
Rights and permissions
About this article
Cite this article
Soo Choi, H., Liu, W., Misra, P. et al. Renal clearance of quantum dots. Nat Biotechnol 25, 1165–1170 (2007). https://doi.org/10.1038/nbt1340
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1340
This article is cited by
-
A renal clearable fluorogenic probe for in vivo β-galactosidase activity detection during aging and senolysis
Nature Communications (2024)
-
Physiological principles underlying the kidney targeting of renal nanomedicines
Nature Reviews Nephrology (2024)
-
Active targeting schemes for nano-drug delivery systems in osteosarcoma therapeutics
Journal of Nanobiotechnology (2023)
-
Multifunctional gold nanoparticles for osteoporosis: synthesis, mechanism and therapeutic applications
Journal of Translational Medicine (2023)
-
Effect of sp3/sp2 carbon ratio and hydrodynamic size on the biodistribution kinetics of nanodiamonds in mice via intravenous injection
Particle and Fibre Toxicology (2023)