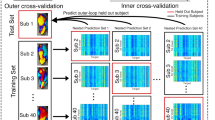
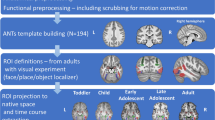
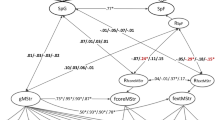
Abstract
A fundamental assumption in neuroscience is that brain structure determines function. Accordingly, functionally distinct regions of cortex should be structurally distinct in their connections to other areas. We tested this hypothesis in relation to face selectivity in the fusiform gyrus. By using only structural connectivity, as measured through diffusion-weighted imaging, we were able to predict functional activation to faces in the fusiform gyrus. These predictions outperformed two control models and a standard group-average benchmark. The structure–function relationship discovered from the initial participants was highly robust in predicting activation in a second group of participants, despite differences in acquisition parameters and stimuli. This approach can thus reliably estimate activation in participants who cannot perform functional imaging tasks and is an alternative to group-activation maps. Additionally, we identified cortical regions whose connectivity was highly influential in predicting face selectivity within the fusiform, suggesting a possible mechanistic architecture underlying face processing in humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Johansen-Berg, H. et al. Changes in connectivity profiles define functionally distinct regions in human medial frontal cortex. Proc. Natl. Acad. Sci. USA 101, 13335–13340 (2004).
Passingham, R.E., Stephan, K.E. & Kotter, R. The anatomical basis of functional localization in the cortex. Nat. Rev. Neurosci. 3, 606–616 (2002).
Haxby, J.V. et al. The effect of face inversion on activity in human neural systems for face and object perception. Neuron 22, 189–199 (1999).
Tsao, D.Y., Schweers, N., Moeller, S. & Freiwald, W.A. Patches of face-selective cortex in the macaque frontal lobe. Nat. Neurosci. 11, 877–879 (2008).
Perrett, D.I., Hietanen, J.K., Oram, M.W. & Benson, P.J. Organization and functions of cells responsive to faces in the temporal cortex. Phil. Trans. R. Soc. Lond. B 335, 23–30 (1992).
Tsao, D.Y., Freiwald, W.A., Tootell, R.B. & Livingstone, M.S. A cortical region consisting entirely of face-selective cells. Science 311, 670–674 (2006).
Moeller, S., Freiwald, W.A. & Tsao, D.Y. Patches with links: a unified system for processing faces in the macaque temporal lobe. Science 320, 1355–1359 (2008).
Kanwisher, N., McDermott, J. & Chun, M.M. The fusiform face area: a module in human extrastriate cortex specialized for face perception. J. Neurosci. 17, 4302–4311 (1997).
Kanwisher, N., Stanley, D. & Harris, A. The fusiform face area is selective for faces not animals. Neuroreport 10, 183–187 (1999).
Epstein, R. & Kanwisher, N. A cortical representation of the local visual environment. Nature 392, 598–601 (1998).
Barton, J.J., Press, D.Z., Keenan, J.P. & O'Connor, M. Lesions of the fusiform face area impair perception of facial configuration in prosopagnosia. Neurology 58, 71–78 (2002).
Pitcher, D., Walsh, V., Yovel, G. & Duchaine, B. TMS evidence for the involvement of the right occipital face area in early face processing. Curr. Biol. 17, 1568–1573 (2007).
McNeil, J.E. & Warrington, E.K. Prosopagnosia: a face-specific disorder. Q. J. Exp. Psychol. A 46, 1–10 (1993).
Landis, T., Cummings, J.L., Christen, L., Bogen, J.E. & Imhof, H.G. Are unilateral right posterior cerebral lesions sufficient to cause prosopagnosia? Clinical and radiological findings in six additional patients. Cortex 22, 243–252 (1986).
McCarthy, G., Puce, A., Gore, J.C. & Allison, T. Face-specific processing in the human fusiform gyrus. J. Cogn. Neurosci. 9, 605–610 (1997).
Behrens, T.E. et al. Non-invasive mapping of connections between human thalamus and cortex using diffusion imaging. Nat. Neurosci. 6, 750–757 (2003).
Behrens, T.E. et al. Characterization and propagation of uncertainty in diffusion-weighted MR imaging. Magn. Reson. Med. 50, 1077–1088 (2003).
Catani, M., Jones, D.K., Donato, R. & Ffytche, D.H. Occipito-temporal connections in the human brain. Brain 126, 2093–2107 (2003).
Seltzer, B. & Pandya, D.N. Parietal, temporal, and occipital projections to cortex of the superior temporal sulcus in the rhesus monkey: a retrograde tracer study. J. Comp. Neurol. 343, 445–463 (1994).
Gloor, P. The Temporal Lobe and Limbic System (Oxford Univ. Press, New York, 1997).
Gholipour, A., Kehtarnavaz, N., Briggs, R., Devous, M. & Gopinath, K. Brain functional localization: a survey of image registration techniques. IEEE Trans. Med. Imaging 26, 427–451 (2007).
Hastie, T., Tibshirani, R. & Friedman, J.H. The Elements of Statistical Learning: Data Mining, Inference, and Prediction (Springer, New York, 2009).
Golland, P. & Fischl, B. Permutation tests for classification: towards statistical significance in image-based studies. Inf. Process. Med. Imaging 18, 330–341 (2003).
Hilgetag, C.C. & Kaiser, M. Clustered organization of cortical connectivity. Neuroinformatics 2, 353–360 (2004).
Sporns, O. & Zwi, J.D. The small world of the cerebral cortex. Neuroinformatics 2, 145–162 (2004).
Saxe, R., Moran, J.M., Scholz, J. & Gabrieli, J. Overlapping and non-overlapping brain regions for theory of mind and self reflection in individual subjects. Soc. Cogn. Affect. Neurosci. 1, 229–234 (2006).
Hinds, O.P. et al. Accurate prediction of V1 location from cortical folds in a surface coordinate system. Neuroimage 39, 1585–1599 (2008).
Annese, J., Gazzaniga, M. & Toga, A. Localization of the human cortical visual area MT based on computer aided histological analysis. Cereb. Cortex 15, 1044–1053 (2005).
Dickerson, B.C. et al. Detection of cortical thickness correlates of cognitive performance: reliability across MRI scan sessions, scanners, and field strengths. Neuroimage 39, 10–18 (2008).
Ishai, A. Let's face it: it's a cortical network. Neuroimage 40, 415–419 (2008).
Kanwisher, N. & Yovel, G. The fusiform face area: a cortical region specialized for the perception of faces. Phil. Trans. R. Soc. Lond. B 361, 2109–2128 (2006).
Epstein, R.A. Parahippocampal and retrosplenial contributions to human spatial navigation. Trends Cogn. Sci. 12, 388–396 (2008).
Sewards, T.V. Neural structures and mechanisms involved in scene recognition: a review and interpretation. Neuropsychologia 49, 277–298 (2011).
Haxby, J.V., Hoffman, E.A. & Gobbini, M.I. The distributed human neural system for face perception. Trends Cogn. Sci. 4, 223–233 (2000).
Schmahmann, J.D. & Pandya, D.N. Course of the fiber pathways to pons from parasensory association areas in the rhesus monkey. J. Comp. Neurol. 326, 159–179 (1992).
Schmahmann, J.D. & Pandya, D.N. Prelunate, occipitotemporal, and parahippocampal projections to the basis pontis in rhesus monkey. J. Comp. Neurol. 337, 94–112 (1993).
Glickstein, M. et al. Visual pontocerebellar projections in the macaque. J. Comp. Neurol. 349, 51–72 (1994).
O'Reilly, J.X., Beckmann, C.F., Tomassini, V., Ramnani, N. & Johansen-Berg, H. Distinct and overlapping functional zones in the cerebellum defined by resting state functional connectivity. Cereb. Cortex 20, 953–965 (2010).
Dauguet, J. et al. Comparison of fiber tracts derived from in-vivo DTI tractography with 3D histological neural tract tracer reconstruction on a macaque brain. Neuroimage 37, 530–538 (2007).
Peled, S., Berezovskii, V., Hendrickson, P., Born, R. & Westin, C. Histological validation of DTI using WGA-HRP in a macaque. Proc. Intl. Soc. Mag. Reson. Med. 13, 1323 (2005).
Thomas, C. et al. Reduced structural connectivity in ventral visual cortex in congenital prosopagnosia. Nat. Neurosci. 12, 29–31 (2009).
Reese, T.G., Heid, O., Weisskoff, R.M. & Wedeen, V.J. Reduction of eddy-current-induced distortion in diffusion MRI using a twice-refocused spin echo. Magn. Reson. Med. 49, 177–182 (2003).
Oliva, A. & Torralba, A. Modeling the shape of the scene: a holistic representation of the spatial envelope. Int. J. Comput. Vis. 42, 145–175 (2001).
Dale, A.M. Optimal experimental design for event-related fMRI. Hum. Brain Mapp. 8, 109–114 (1999).
Pitcher, D., Dilks, D.D., Saxe, R.R., Triantafyllou, C. & Kanwisher, N. Differential selectivity for dynamic versus static information in face-selective cortical regions. Neuroimage 56, 2356–2363 (2011).
Fischl, B. et al. Automatically parcellating the human cerebral cortex. Cereb. Cortex 14, 11–22 (2004).
Fischl, B. et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33, 341–355 (2002).
Desikan, R.S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31, 968–980 (2006).
Behrens, T.E., Berg, H.J., Jbabdi, S., Rushworth, M.F. & Woolrich, M.W. Probabilistic diffusion tractography with multiple fibre orientations: what can we gain? Neuroimage 34, 144–155 (2007).
Acknowledgements
We thank N. Kanwisher, S. Ghosh, F. Polli and the Athinoula A. Martinos Imaging Center at McGovern Institute for Brain Research, Massachusetts Institute of Technology. This work was supported by US Public Health Service DA023427, US National Institute of Mental Health F32 MH084488, US National Eye Institute T32 EY013935, the Poitras Center for Affective Disorders Research, the Simons Foundation and the Ellison Medical Foundation.
Author information
Authors and Affiliations
Contributions
Z.M.S. and D.E.O. designed and performed experiments, analyzed data and wrote the manuscript. K.K. designed and performed experiments. G.R. performed experiments and analyzed data. J.D.E.G. and R.R.S. designed experiments and helped write the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Table 1, Supplementary Results and Supplementary Discussion (PDF 803 kb)
Rights and permissions
About this article
Cite this article
Saygin, Z., Osher, D., Koldewyn, K. et al. Anatomical connectivity patterns predict face selectivity in the fusiform gyrus. Nat Neurosci 15, 321–327 (2012). https://doi.org/10.1038/nn.3001
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3001
This article is cited by
-
How do Robot Touch Characteristics Impact Users’ Emotional Responses: Evidence from ECG and fNIRS
International Journal of Social Robotics (2024)
-
Accurate localization and coactivation profiles of the frontal eye field and inferior frontal junction: an ALE and MACM fMRI meta-analysis
Brain Structure and Function (2023)
-
Ramp-shaped neural tuning supports graded population-level representation of the object-to-scene continuum
Scientific Reports (2022)
-
Establishing the functional relevancy of white matter connections in the visual system and beyond
Brain Structure and Function (2022)
-
Development of white matter tracts between and within the dorsal and ventral streams
Brain Structure and Function (2022)