Key Points
-
The dominant view in current theories of face perception is that facial identity (recognizing who a person is) and facial expression (interpreting their moods and feelings) are processed by distinct parallel visual routes. Although there is considerable evidence to support the independent coding of identity and expression, it is not clear whether the idea of distinct parallel visual routes provides the best fit to the data.
-
We conclude that there is clear evidence for some separation between the coding of facial identity and expression, the concept of independent visual pathways is not strongly supported. The data are consistent with other potential frameworks that deserve to be more fully explored.
-
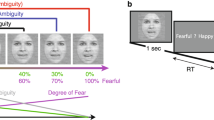
One alternative framework derives from image-based analysis of faces using principal component analysis (PCA). This shows that the independent perception of facial identity and facial expression can be modelled within a single representational framework in which some dimensions (principal components) code facial identity, some code facial expressions and others code both. PCA therefore indicates that the dissociation of identity and expression might be partial rather than absolute.
-
We also focus on Haxby and colleagues' observations that facial expressions and other 'changeable' facial cues (such as lipspeech and gaze) are associated with the inferior occipital gyrus and superior temporal sulcus (STS), whereas 'invariant' facial cues (such as facial identity) are associated with the inferior occipital gyrus and lateral fusiform gyrus. This distinction begs more fundamental questions, such as why are facial characteristics divided in this manner and why is the STS more interested in facial expressions, lipspeech and gaze?
-
One potential explanation lies in the fact that the STS is not only sensitive to changeable facial characteristics, but also to other perceptual dimensions that are inherently linked with them (such as their associated vocalizations and dynamic information). There is evidence that the STS might be involved in the integration of these different channels. Consequently, we propose that the prominent role of the STS in coding changeable facial characteristics might reflect an increased reliance on integrative mechanisms for interpreting changeable social signals.
-
In summary, an approach to face perception that emphasizes the different physical properties and information-processing demands (such as reliance on integrative mechanisms) of different facial characteristics has considerable value. This differs from the classic approach, which has tended to emphasize distinctions based mainly on informational content (for example, identity versus expression).
Abstract
Faces convey a wealth of social signals. A dominant view in face-perception research has been that the recognition of facial identity and facial expression involves separable visual pathways at the functional and neural levels, and data from experimental, neuropsychological, functional imaging and cell-recording studies are commonly interpreted within this framework. However, the existing evidence supports this model less strongly than is often assumed. Alongside this two-pathway framework, other possible models of facial identity and expression recognition, including one that has emerged from principal component analysis techniques, should be considered.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bruce, V. & Young, A. W. Understanding face recognition. Br. J. Psychol. 77, 305–327 (1986). A theoretical paper that proposes a multiroute model of face perception in which different facial attributes are processed by distinct functional routes.
Haxby, J. V., Hoffman, E. A. & Gobbini, M. I. The distributed human neural system for face perception. Trends Cogn. Sci. 4, 223–233 (2000). Presents a neural model of face perception that comprises 'core' and 'extended' systems. The core system differentiates mechanisms for coding changeable facial properties and mechanisms coding invariant facial properties. The extended system includes neural regions that are involved in semantics, language, emotion and attention, which support the recognition of different facial characteristics.
Bruce, V. Influences of familiarity on the processing of faces. Perception 15, 387–397 (1986).
Campbell, R., Brooks, B., de Haan, E. & Roberts, T. Dissociating face processing skills: decisions about lip-read speech, expression and identity. Q. J. Exp. Psychol. A 49A, 295–314 (1996).
Young, A. W., McWeeny, K. H., Hay, D. C. & Ellis, A. W. Matching familiar and unfamiliar faces on identity and expression. Psychol. Res. 48, 63–68 (1986).
Calder, A. J., Young, A. W., Keane, J. & Dean, M. Configural information in facial expression perception. J. Exp. Psychol. Hum. Percept. Perform. 26, 527–551 (2000).
Etcoff, N. L. Selective attention to facial identity and facial emotion. Neuropsychologia 22, 281–295 (1984).
Young, A. W., Newcombe, F., de Haan, E. H. F., Small, M. & Hay, D. C. Face perception after brain injury: selective impairments affecting identity and expression. Brain 116, 941–959 (1993).
Bruyer, R. et al. A case of prosopagnosia with some preserved covert remembrance of familiar faces. Brain Cogn. 2, 257–284 (1983).
Tranel, D., Damasio, A. R. & Damasio, H. Intact recognition of facial expression, gender, and age in patients with impaired recognition of face identity. Neurology 38, 690–696 (1988).
Hornak, J., Rolls, E. T. & Wade, D. Face and voice expression identification in patients with emotion and behavioural changes following ventral frontal lobe damage. Neuropsychologia 34, 247–261 (1996).
Hasselmo, M. E., Rolls, E. T. & Baylis, G. C. The role of expression and identity in face-selective responses of neurons in the temporal visual cortex of the monkey. Behav. Brain Res. 32, 203–218 (1989).
George, M. S. et al. Brain-regions involved in recognizing facial emotion or identity: an oxygen-15 PET study. J. Neuropsychiatry Clin. Neurosci. 5, 384–394 (1993).
Sergent, J., Ohta, S., MacDonald, B. & Zuck, E. Segregated processing of identity and emotion in the human brain: a PET study. Vis. Cogn. 1, 349–369 (1994).
Winston, J. S., Henson, R. N. A., Fine-Goulden, M. R. & Dolan, R. J. fMRI-adaptation reveals dissociable neural representations of identity and expression in face perception. J. Neurophysiol. 92, 1830–1839 (2004).
Parry, F. M., Young, A. W., Saul, J. S. M. & Moss, A. Dissociable face processing impairments after brain injury. J. Clin. Exp. Neuropsychol. 13, 545–558 (1990).
Shuttleworth, E. C. J., Syring, V. & Allen, N. Further observations on the nature of prosopagnosia. Brain Cogn. 1, 307–322 (1982).
Hanley, J. R., Pearson, N. & Young, A. W. Impaired memory for new visual forms. Brain 113, 1131–1148 (1990). Reports the first detailed case study of a patient who showed impaired learning of faces (prosopamnesia) and of visual forms in general after a brain injury.
Tippet, L. J., Milller, L. A. & Farah, M. J. Prosopamnesia: a selective impairment in face learning. Cogn. Neuropsychol. 17, 241–255 (2000).
Hanley, J. R., Young, A. W. & Pearson, N. Defective recognition of familiar people. Cogn. Neuropsychol. 6, 179–210 (1989).
Evans, J. J., Heggs, A. J., Antoun, N. & Hodges, J. R. Progressive prosopagnosia associated with selective right temporal lobe atrophy: a new syndrome? Brain 118, 1–13 (1995).
Ellis, A. W., Young, A. W. & Critchley, E. M. R. Loss of memory for people following temporal lobe damage. Brain 112, 1469–1483 (1989). Reports the first detailed case study of a patient who showed impaired access to person-specific semantic information from several modalities.
Malone, D. R., Morris, H. H., Kay, M. C. & Levin, H. S. Prosopagnosia: a double dissociation between the recognition of familiar and unfamiliar faces. J. Neurol. Neurosurg. Psychiatry 45, 820–822 (1982).
Ekman, P. & Friesen, W. V. Pictures of Facial Affect (Consulting Psychologists, Palo Alto, California, 1976).
Sprengelmeyer, R. et al. Loss of disgust: perception of faces and emotions in Huntington's disease. Brain 119, 1647–1665 (1996).
Calder, A. J., Keane, J., Manes, F., Antoun, N. & Young, A. W. Impaired recognition and experience of disgust following brain injury. Nature Neurosci. 3, 1077–1078 (2000).
Rapcsak, S. Z. et al. Fear recognition deficits after focal brain damage: a cautionary note. Neurology 54, 575–581 (2000).
Bowers, D., Bauer, R. M., Coslett, H. B. & Heilman, K. M. Processing of faces by patients with unilateral hemisphere lesions. 1. Dissociation between judgements of facial affect and facial identity. Brain Cogn. 4, 258–272 (1985).
Shallice, T. From Neuropsychology to Mental Structure (Cambridge Univ. Press, Cambridge, 1988).
Duchaine, B. C., Paerker, H. & Nakayama, K. Normal recognition of emotion in a prosopagnosic patient. Perception 32, 827–839 (2003).
Bishop, D. V. M. Cognitive neuropsychology and developmental disorders: uncomfortable bedfellows. Q. J. Exp. Psychol. A 50A, 899–923 (1997).
Karmiloff-Smith, A., Scerif, G. & Ansari, D. Double dissociations in developmental disorders? Theoretically misconceived, empirically dubious. Cortex 39, 161–163 (2003).
Turk, M. & Pentland, A. Eigenfaces for recognition. J. Cogn. Neurosci. 3, 71–86 (1991).
Burton, A. M., Bruce, V. & Hancock, P. J. B. From pixels to people: a model of familiar face recognition. Cogn. Sci. 23, 1–31 (1999). Presents a model of facial identity recognition in which the front end is implemented as a PCA, and the subsequent cognitive processes are implemented as an interactive activation and competition architecture.
Hancock, P. J. B., Burton, A. M. & Bruce, V. Face processing: human perception and principal components analysis. Mem. Cognit. 24, 21–40 (1996).
Hancock, P. J. B., Bruce, V. & Burton, A. M. A comparison of two computer-based face identification systems with human perception of faces. Vision Res. 38, 2277–2288 (1998).
O'Toole, A. J. et al. The perception of face gender: the role of stimulus structure in recognition and classification. Mem. Cognit. 26, 146–160 (1998).
O'Toole, A. J., Abdi, H., Deffenbacher, K. A. & Valentin, D. Low-dimensional representation of faces in higher dimensions of the face space. J. Opt. Soc. Am. 10, 405–411 (1993).
O'Toole, A. J., Deffenbacher, K. A., Valentin, D. & Abdi, H. Structural aspects of face recognition and the other race effect. Mem. Cognit. 22, 208–224 (1994).
Padgett, C. & Cottrell, G. in Proceedings of the 2nd Joint Symposium on Neural Computation Vol. 5, 91–101 (La Jolla, Univ. California, San Diego, 1995).
Cottrell, G. W., Branson, K. M. & Calder, A. J. in 24th Annual Meeting of the Cognitive Science Society (Fairfax, Virginia, 2002).
Calder, A. J., Burton, A. M., Miller, P., Young, A. W. & Akamatsu, S. A principal component analysis of facial expressions. Vision Res. 41, 1179–1208 (2001). Provides evidence that a PCA of the Ekman and Friesen facial expressions (reference 24) generates statistical dissociation between PCs that code identity and expression.
Costen, N., Craw, I. & Akamatsu, S. in British Machine Vision Association (Scottish Chapter) Meeting (British Machine Vision Association, Glasgow, 1995).
Deffenbacher, K. A., Vetter, T., Johanson, J. & O'Toole, A. J. Facial aging, attractiveness, and distinctiveness. Perception 27, 1233–1243 (1998).
Schweinberger, S. R. & Soukup, G. R. Asymmetric relationships among perceptions of facial identity, emotion, and facial speech. J. Exp. Psychol. Hum. Percept. Perform. 24, 1748–1765 (1998).
Narumoto, J., Okada, T., Sadato, N., Fukui, K. & Yonekura, Y. Attention to emotion modulates fMRI activity in human right superior temporal sulcus. Cogn. Brain Res. 12, 225–231 (2001).
Sugase, Y., Yamane, S., Ueno, S. & Kawano, K. Global and fine information coded by single neurons in the temporal visual cortex. Nature 400, 869–873 (1999).
Phillips, M. L. et al. Neural responses to facial and vocal expressions of fear and disgust. Proc. R. Soc. Lond. B 265, 1809–1817 (1998).
Morris, J. S. et al. A neuromodulatory role for the human amygdala in processing emotional facial expressions. Brain 121, 47–57 (1998).
Lewis, S. et al. Visual processing of facial affect. Neuroreport 14, 1841–1845 (2003).
Streit, M. et al. Neurophysiological correlates of the recognition of facial expressions of emotion as revealed by magnetoencephalography. Cogn. Brain Res. 7, 481–491 (1999).
Kanwisher, N., McDermott, J. & Chun, M. M. The fusiform face area: a module in human extrastriate cortex specialized for face perception. J. Neurosci. 17, 4302–4311 (1997).
McCarthy, G., Puce, A., Core, J. C. & Allison, T. Face-specific processing in the human fusiform gyrus. J. Cogn. Neurosci. 9, 605–610 (1997).
Henson, R., Shallice, T. & Dolan, R. Neuroimaging evidence for dissociable forms of repetition priming. Science 287, 1269–1272 (2000).
Perrett, D. I. et al. Neurones responsive to faces in the temporal cortex: studies of functional organization, sensitivity to identity and relation to perception. Hum. Neurobiol. 3, 197–208 (1984).
Perrett, D. I., Hietanen, J. K., Oram, M. W. & Benson, P. J. Organization and functions of cells responsive to faces in the temporal cortex. Phil. Trans. R. Soc. Lond. B B335, 23–30 (1992).
Young, M. P. & Yamane, S. Sparse population coding of faces in the inferotemporal cortex. Science 256, 1327–1331 (1992). Shows that face-responsive cells in the inferotemporal cortex of monkeys code visual properties, whereas face-responsive cells in the STS code nonvisual properties, such as familiarity or social status.
Yamane, S., Kaji, S. & Kawano, K. What facial features activate face neurones in the inferotemporal cortex in the monkey? Exp. Brain Res. 73, 209–214 (1988).
Rolls, E. T. & Tovee, M. J. Sparseness of the neuronal representation of stimuli in the primate temporal visual cortex. J. Neurophysiol. 73, 713–726 (1995).
Seltzer, B. & Pandya, D. N. Afferent cortical connections and architectonics of the superior temporal sulcus and surrounding cortex in the rhesus monkey. Brain Res. 149, 1–24 (1978).
Bruce, C., Desimone, R. & Gross, C. G. Visual properties of neurons in a polysensory area in superior temporal cortex. J. Neurophysiol. 46, 369–384 (1981).
Adolphs, R., Tranel, D., Damasio, H. & Damasio, A. Impaired recognition of emotion in facial expressions following bilateral damage to the human amygdala. Nature 372, 669–672 (1994).
Sprengelmeyer, R. et al. Knowing no fear. Proc. R. Soc. Lond. B 266, 2451–2456 (1999).
Calder, A. J. et al. Facial emotion recognition after bilateral amygdala damage: differentially severe impairment of fear. Cogn. Neuropsychol. 13, 699–745 (1996).
Calder, A. J., Lawrence, A. D. & Young, A. W. The neuropsychology of fear and loathing. Nature Rev. Neurosci. 2, 352–363 (2001).
Sprengelmeyer, R. et al. Disgust implicated in obsessive-compulsive disorder. Proc. R. Soc. Lond. B 264, 1767–1773 (1997).
Calder, A. J., Keane, J., Lawrence, A. D. & Manes, F. Impaired recognition of anger following damage to the ventral striatum. Brain 127, 1958–1969 (2004).
Lawrence, A. D., Calder, A. J., McGowan, S. W. & Grasby, P. M. Selective disruption of the recognition of facial expressions of anger. Neuroreport 13, 881–884 (2002).
Krolak-Salmon, P. et al. A specific response to disgust modulated by attention in human ventral anterior insula. Ann. Neurol. 53, 446–453 (2003).
Morris, J. S. et al. A differential neural response in the human amygdala to fearful and happy facial expressions. Nature 383, 812–815 (1996).
Breiter, H. C. et al. Response and habituation of the human amygdala during visual processing of facial expression. Neuron 17, 875–887 (1996).
Phillips, M. L. et al. A specific neural substrate for perceiving facial expressions of disgust. Nature 389, 495–498 (1997).
Lawrence, A. D. & Calder, A. J. in Emotion, Evolution, and Rationality (eds Evans, D. & Cruse, P.) 15–47 (Oxford Univ. Press, Oxford, 2003).
Scott, S. K. et al. Impaired auditory recognition of fear and anger following bilateral amygdala lesions. Nature 385, 254–257 (1997).
Adolphs, R. & Tranel, D. Intact recognition of emotional prosody following amygdala damage. Neuropsychologia 37, 1285–1292 (1999).
Anderson, A. K. & Phelps, E. A. Intact recognition of vocal expressions of fear following bilateral lesions of the human amygdala. Neuroreport 9, 3607–3613 (1998).
Wicker, B. et al. Both of us disgusted in my insula: the common neural basis of seeing and feeling disgust. Neuron 40, 655–664 (2003).
Gallese, V., Keysers, C. & Rizzolatti, G. A unifying view of the basis of social cognition. Trends Cogn. Sci. 8, 396–403 (2004).
Goldman, A. I. & Sekhar Sripada, C. Simulationist models of face-based emotion recognition. Cognition 94, 193–213 (2005).
Keane, J., Calder, A. J., Hodges, J. R. & Young, A. W. Face and emotion processing in frontal variant frontotemporal dementia. Neuropsychologia 40, 655–665 (2002).
Adolphs, R., Tranel, D. & Damasio, H. Neural systems for recognizing emotion from prosody. Emotion 2, 23–51 (2002).
Adolphs, R., Damasio, H., Tranel, D., Cooper, G. & Damasio, A. R. A role for somatosensory cortices in the visual recognition of emotion as revealed by three-dimensional lesion mapping. J. Neurosci. 20, 2683–2690 (2000).
Anderson, A. K. & Phelps, E. A. Lesions of the human amygdala impair enhanced perception of emotionally salient events. Nature 411, 305–309 (2001).
Anderson, A. K., Spencer, D. D., Fulbright, R. K. & Phelps, E. A. Contribution of the anteromedial temporal lobes to the evaluation of facial emotion. Neuropsychology 14, 526–536 (2000).
Labar, K. S., LeDoux, J. E., Spencer, D. D. & Phelps, E. A. Impaired fear conditioning following unilateral temporal lobectomy in humans. J. Neurosci. 15, 6846–6855 (1995).
LaBar, K. S. & Phelps, E. A. Arousal-mediated memory consolidation: role of the medial temporal lobe in humans. Psychol. Sci. 9, 490–493 (1998).
Phelps, E. A. et al. Specifying the contributions of the human amygdala to emotional memory: a case study. Neurocase 4, 527–540 (1998).
Adolphs, R., Cahill, L., Schul, R. & Babinsky, R. Impaired declarative memory for emotional material following bilateral amygdala damage in humans. Learn. Mem. 4, 291–300 (1997).
Bechara, A. et al. Double dissociation of conditioning and declarative knowledge relative to the amygdala and hippocampus in humans. Science 269, 1115–1118 (1995).
Barraclough, N. E., Xiao, D., Baker, C. I., Oram, M. W. & Perrett, D. I. Integration of visual and auditory information by superior temporal sulcus neurons responsive to the sight of actions. J. Cogn. Neurosci. 17, 377–391 (2005).
Dolan, R. J., Morris, J. S. & de Gelder, B. Crossmodal binding of fear in voice and face. Proc. Natl Acad. Sci. USA 98, 10006–10010 (2001).
Calvert, G. A., Campbell, R. & Brammer, M. J. Evidence from functional magnetic resonance imaging of crossmodal binding in human heteromodal cortex. Curr. Biol. 10, 649–657 (2000). Provides evidence from fMRI that the STS is involved in the integration of seen and heard speech.
Beauchamp, M. S., Argall, B. D., Bodurka, J., Duyn, J. H. & Martin, A. Unravelling multisensory integration: patchy organization within human STS multisensory cortex. Nature Neurosci. 7, 1190–1192 (2004).
Campbell, R. et al. Cortical substrates for the perception of face actions: an fMRI study of the specificity of activation for seen speech and for meaningless lower-face acts (gurning). Cogn. Brain Res. 12, 233–243 (2001).
Belin, P., Zatorre, R. J., Lafaille, P., Ahad, P. & Pike, B. Voice-selective areas in human auditory cortex. Nature 403, 309–312 (2000).
Desimone, R. & Gross, C. G. Visual areas in the temporal cortex of the macaque. Brain Res. 178, 363–380 (1979).
Oram, M. W. & Perrett, D. I. Integration of form and motion in the anterior superior temporal polysensory area (STPa) of the macaque monkey. J. Neurophysiol. 76, 109–129 (1996). Reports evidence from cell recording in monkeys that the STS is involved in the integration of visual form and motion components of biological motion.
McGurk, H. & MacDonald, J. Hearing lips and seeing voices. Nature 264, 746–748 (1976).
de Gelder, B. & Vroomen, J. The perception of emotions by ear and by eye. Cogn. Emotion 14, 289–311 (2000).
Rosenblum, L. D., Johnson, J. A. & Saldana, H. M. Point-light facial displays enhance comprehension of speech in noise. J. Speech Hear. Res. 39, 1159–1170 (1996).
Tartter, V. C. & Knowlton, K. C. Perception of sign language from an array of 27 moving spots. Nature 289, 676–678 (1981).
Puce, A., Allison, T., Bentin, S., Gore, J. C. & McCarthy, G. Temporal cortex activation in humans viewing eye and mouth movements. J. Neurosci. 18, 2188–2199 (1998).
Hoffman, E. A. & Haxby, J. V. Distinct representations of eye gaze and identity in the distributed human neural system for face perception. Nature Neurosci. 3, 80–84 (2000).
Langton, S. R. H. The mutual influence of gaze and head orientation in the analysis of social attention direction. Q. J. Exp. Psychol. A 53, 825–845 (2000).
Langton, S. R. H., Honeyman, H. & Tessler, E. The influence of head contour and nose angle on the perception of eye-gaze direction. Percept. Psychophys. 66, 752–771 (2004).
Langton, S. R. H. & Bruce, V. You must see the point: automatic processing of cues to the direction of social attention. J. Exp. Psychol. Hum. Percept. Perform. 26, 747–757 (2000).
Adams, R. B. & Kleck, R. E. Perceived gaze direction and the processing of facial displays of emotion. Psychol. Sci. 14, 644–647 (2003). Presents evidence that gaze direction affects the perception of emotion from faces.
Hanley, J. R., Smith, S. T. & Hadfield, J. I recognise you but I can't place you: an investigation of familiar-only experiences during tests of voice and face recognition. Q. J. Exp. Psychol. A 51A, 179–195 (1998).
Bruce, V. & Valentine, T. in Practical Aspects of Memory: Current Research and Issues Vol. 1 (eds Gruneberg, M. M., Morris, P. E. & Sykes, R. N.) 169–174 (Wiley, Chichester, 1988).
Bonda, E., Petrides, M., Ostry, D. & Evans, A. Specific involvement of human parietal systems and the amygdala in the perception of biological motion. J. Neurosci. 16, 3737–3744 (1996).
Grossman, E. et al. Brain areas involved in perception of biological motion. J. Cogn. Neurosci. 12, 711–720 (2000).
Castelli, F., Happe, F., Frith, U. & Frith, C. Movement and mind: a functional imaging study of perception and interpretation of complex intentional movement patterns. Neuroimage 12, 314–325 (2000).
Allison, T., Puce, A. & McCarthy, G. Social perception from visual cues: role of the STS region. Trends Cogn. Sci. 4, 267–278 (2000).
Beauchamp, M. S., Lee, K. E., Haxby, J. V. & Martin, A. fMRI responses to video and point-light displays of moving and manipulable objects. J. Cogn. Neurosci. 15, 991–1001 (2003).
Calvert, G. A. et al. Activation of auditory cortex during silent lipreading. Science 276, 593–596 (1997).
Campbell, R., Landis, T. & Regard, M. Face recognition and lipreading: a neurological dissociation. Brain 109, 509–521 (1986).
Adams, R. B. & Kleck, R. E. The effects of direct and averted gaze on the perception of facial communicated emotion. Emotion 5, 3–11 (2005).
Adams, R. B., Gordon, H. L., Baird, A. A., Ambady, N. & Kleck, R. E. Gaze differentially modulates amygdala sensitivity to anger and fear faces. Science 300, 1536 (2003).
O'Toole, A. J., Roark, D. A. & Abdi, H. Recognizing moving faces: a psychological and neural synthesis. Trends Cogn. Sci. 6, 261–266 (2002).
Simons, D. J. & Levin, D. T. Failure to detect changes to people during a real-world interaction. Psychol. Bull. Rev. 5, 644–649 (1998).
Dimberg, U. Facial reactions for facial expressions. J. Psychophys. 19, 643–647 (1982).
Watkins, K. E., Strafella, A. P. & Paus, T. Seeing and hearing speech excites the motor system involved in speech production. Neuropsychologia 41, 989–994 (2003).
Langton, S. R. H. & Bruce, V. Reflexive visual orienting in response to the social attention of others. Vis. Cogn. 6, 541–567 (1999).
Driver, J. et al. Gaze perception triggers reflexive visuospatial orienting. Vis. Cogn. 6, 509–540 (1999).
Friesen, C. K. & Kingstone, A. The eyes have it! Reflexive orienting is triggered by nonpredictive gaze. Psychol. Bull. Rev. 5, 490–495 (1998).
Acknowledgements
We would like to thank B. Cox for assistance in preparing the figures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- FUNCTIONAL ACCOUNT
-
A theoretical framework that is based on the information-processing characteristics of a set of cognitive subsystems rather than their underlying neural mechanisms
- PROSOPAGNOSIA
-
A visual agnosia that is largely restricted to face recognition, but leaves intact recognition of personal identity from other identifying cues, such as voices and names. Prosopagnosia is observed after bilateral and, less frequently, unilateral lesions of the inferior occipitotemporal cortex.
- ACQUIRED NEUROPSYCHOLOGICAL DISORDERS
-
Cognitive impairments that follow neurological damage to previously healthy individuals who have no known genetic or developmental disorders.
- PRINCIPAL COMPONENT ANALYSIS
-
(PCA). A statistical technique that been applied to the analysis of faces. Facial images, which are originally described in terms of a large number of variables (for example, the greyscale values of individual pixels), are recoded in relation to a smaller set of basis vectors (principal components) by identifying correlations between sets of pixels across the entire set of facial images.
- POPULATION CODING
-
The idea that objects, such as faces, are coded as distributed patterns of activity across neuronal populations, so that cells show broadly graded responses to a single stimulus, as opposed to an 'all-or-none'-type response pattern.
- GRANDMOTHER CELL
-
A hypothetical cell that responds specifically to a single face (such as one's grandmother). Although no such highly specialized cell has been found so far, the temporal cortex contains cells that respond preferentially to faces or hands, and some are maximally (although not exclusively) responsive to a particular persons' face. These cells are sometimes referred to as 'grandmother cells'.
- HEIDER-SIMMEL-LIKE ANIMATIONS
-
Short animations that depict the movements of geometric shapes in a manner that is normally perceived as suggestive of social interactions among 'human-like' characters. For example, one shape might be perceived as aggressive and intimidating towards another.
Rights and permissions
About this article
Cite this article
Calder, A., Young, A. Understanding the recognition of facial identity and facial expression. Nat Rev Neurosci 6, 641–651 (2005). https://doi.org/10.1038/nrn1724
Issue Date:
DOI: https://doi.org/10.1038/nrn1724
This article is cited by
-
Domestication constrains the ability of dogs to convey emotions via facial expressions in comparison to their wolf ancestors
Scientific Reports (2024)
-
Realness of face images can be decoded from non-linear modulation of EEG responses
Scientific Reports (2024)
-
To see or not to see: the parallel processing of self-relevance and facial expressions
Cognitive Research: Principles and Implications (2023)
-
Autistic adults exhibit highly precise representations of others’ emotions but a reduced influence of emotion representations on emotion recognition accuracy
Scientific Reports (2023)
-
Toddlers’ sensitivity to dominance traits from faces
Scientific Reports (2023)