Abstract
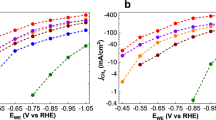
Semiconductors containing transition metal clusters within their crystal lattice have recently been proposed as catalysts for multi-electron transfer reactions. This is because of the expected action of the clusters as reservoirs for electronic charge carriers and sites for cooperative electron transfer at a reasonably constant electrochemical potential1,2. Here we report the first test of the capacity of such materials for oxygen reduction in an acid medium, for operation in fuel cells. The compound Mo4.2Ru1.8Se8, which contains octahedra statistically composed of 4.2 molybdenum and 1.8 ruthenium atoms, turned out to have a catalytic behaviour comparable to platinum, which has so far been the best catalyst for fuel cells. The material costs of this new type of catalyst amount to only a few per cent of those of platinum.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Tributsch, H. J. Photochem. 29, 89–113 (1985).
Tributsch, H. in Modern Aspects of Electrochemistry 17 (eds Bockris, J. O'M., Conway, B. E. & White, R. E.) (Plenum, New York, 1986).
Kühne, H. M. & Tributsch, H. J. electroanalyt. Chem. 201, 263–282 (1986).
Hönle, W., Flack, H. D. & Yvon, K. J. Solid St. Chem. 49, 157–165 (1983).
Tarasevich, A. R., Sadkowski, A. & Yeager, E. in Comprehensive Treatise of Electrochemistry, Vol. 7 (eds Conway, B. E., Bockris, J. O'M., Yeager, E., Khan, S. U. M. & White, R. E.) 301–398 (Plenum, New York, 1983).
Appleby, A. J. Energy 11, 13–94 (1986).
Alonso Vante, N., Jaegermann, W., Hönle, W., Yvon, K. & Tributsch, H. J. Am. chem. Soc. (submitted).
Horkans, J. & Schafer, M. W. J. electrochem. Soc. 124, 1196–1202; 1202–1207 (1977).
Hsueh, K.-L., Gonzalez, E. R. & Srinivasan, S. Electrochim. Acta 12, 691–697 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vante, N., Tributsch, H. Energy conversion catalysis using semiconducting transition metal cluster compounds. Nature 323, 431–432 (1986). https://doi.org/10.1038/323431a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/323431a0
This article is cited by
-
Interfacial water engineering boosts neutral water reduction
Nature Communications (2022)
-
Cobalt-based oxygen electrocatalysts for zinc-air batteries: Recent progress, challenges, and perspectives
Nano Research (2022)
-
Advanced Noncarbon Materials as Catalyst Supports and Non-noble Electrocatalysts for Fuel Cells and Metal–Air Batteries
Electrochemical Energy Reviews (2021)
-
Oxygen Reduction on Methanol-Tolerant Carbon-Supported PtxSy Nanoparticles Prepared by a Single-Step Low-Temperature Method
Electrocatalysis (2019)
-
In situ redox reactions facilitate the assembly of a mixed-valence metal-organic nanocapsule
Nature Communications (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.