Abstract
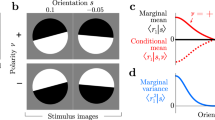
The brain represents sensory and motor variables through the activity of large populations of neurons. It is not understood how the nervous system computes with these population codes, given that individual neurons are noisy and thus unreliable. We focus here on two general types of computation, function approximation and cue integration, as these are powerful enough to handle a range of tasks, including sensorimotor transformations, feature extraction in sensory systems and multisensory integration. We demonstrate that a particular class of neural networks, basis function networks with multidimensional attractors, can perform both types of computation optimally with noisy neurons. Moreover, neurons in the intermediate layers of our model show response properties similar to those observed in several multimodal cortical areas. Thus, basis function networks with multidimensional attractors may be used by the brain to compute efficiently with population codes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pouget, A. & Snyder, L. Computational approaches to sensorimotor transformations. Nat. Neurosci. 3, 1192–1198 (2000).
Poggio, T. A theory of how the brain might work. Cold Spring Harb. Symp. Quant. Biol. 55, 899–910 (1990).
Hubel, D. & Weisel, T. Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J. Physiol. (Lond.) 160, 106–154 (1962).
Douglas, R. J., Koch, C., Mahowald, M., Martin, K. A. & Suarez, H. H. Recurrent excitation in neocortical circuits. Science 269, 981–985 (1995).
Deneve, S., Latham, P. & Pouget, A. Reading population codes: a neural implementation of ideal observers. Nat. Neurosci. 2, 740–745 (1999).
Pouget, A. & Sejnowski, T. Spatial transformations in the parietal cortex using basis functions. J. Cogn. Neurosci. 9, 222–237 (1997).
Westheimer, G. & Tanzman, I. Qualitative depth localization with diplopic images. J. Opt. Soc. Am. 46, 116–117 (1956).
Andersen, R., Essick, G. & Siegel, R. Encoding of spatial location by posterior parietal neurons. Science 230, 456–458 (1985).
Duhamel, J., Bremmer, F., BenHamed, S. & Graf, W. Spatial invariance of visual receptive fields in parietal cortex neurons. Nature 389, 845–848 (1997).
Yuille, A. L. & Bulthoff, H. H. in Perception as Bayesian Inference (eds. Knill, D. C. & Richards, W.) 123–162 (University Press, New York, Cambridge, 1996).
Papoulis, A. Probability, Random Variables, and Stochastic Process (McGraw-Hill, New York, 1991).
Paradiso, M. A theory of the use of visual orientation information which exploits the columnar structure of striate cortex. Biol. Cybern. 58, 35–49 (1988).
Seung, H. & Sompolinsky, H. Simple model for reading neuronal population codes. Proc. Natl. Acad. Sci. USA 90, 10749–10753 (1993).
Tolhurst, D., Movshon, J. & Dean, A. The statistical reliability of signals in single neurons in cat and monkey visual cortex. Vision Res. 23, 775–785 (1982).
Salinas, E. & Abbot, L. Transfer of coded information from sensory to motor networks. J. Neurosci. 15, 6461–6474 (1995).
McAdams, C. J. & Maunsell, J. R. H. Effects of attention on orientation-tuning functions of single neurons in macaque cortical area V4. J. Neurosci. 19, 431–441 (1999).
Braitenberg, V. & Schuz, A. Anatomy of the Cortex (Springer, Berlin, 1991).
Boussaoud, D., Barth, T. & Wise, S. Effects of gaze on apparent visual responses of frontal cortex neurons. Exp. Brain Res. 93, 423–434 (1993).
Bremmer, F., Ilg, U., Thiele, A., Distler, C. & Hoffman, K. Eye position effects in monkey cortex. I: Visual and pursuit-related activity in extrastriate areas MT and MST. J. Neurophysiol. 77, 944–961 (1997).
Trotter, Y. & Celebrini, S. Gaze direction controls response gain in primary visual-cortex neurons. Nature 398, 239–242 (1999).
Jay, M. F. & Sparks, D. L. Sensorimotor integration in the primate superior colliculus: I. Motor convergence. J. Neurophysiol. 57, 22–34 (1987).
Graziano, M., Yap, G. & Gross, C. Coding of visual space by premotor neurons. Science 266, 1054–1057 (1994).
Acknowledgements
A.P and S.D. are supported by grants from NIH (MH57823-05), ONR (N00014-00-1-0642) and a fellowship from the McDonnell-Pew Foundation. P.E.L. is supported by a grant from NIH (MH62447-01). We thank J.-R. Duhamel for discussions.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Deneve, S., Latham, P. & Pouget, A. Efficient computation and cue integration with noisy population codes. Nat Neurosci 4, 826–831 (2001). https://doi.org/10.1038/90541
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/90541
This article is cited by
-
Position- and scale-invariant object-centered spatial localization in monkey frontoparietal cortex dynamically adapts to cognitive demand
Nature Communications (2024)
-
Priority coding in the visual system
Nature Reviews Neuroscience (2022)
-
Advances in the computational understanding of mental illness
Neuropsychopharmacology (2021)
-
Active inference under visuo-proprioceptive conflict: Simulation and empirical results
Scientific Reports (2020)
-
Cortical-like dynamics in recurrent circuits optimized for sampling-based probabilistic inference
Nature Neuroscience (2020)