Abstract
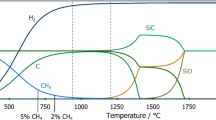
Silicon dioxide (SiO2) is conventionally reduced to silicon by carbothermal reduction, in which the oxygen is removed by a heterogeneous–homogeneous reaction sequence at approximately 1,700 °C. Here we report pinpoint and bulk electrochemical methods for removing oxygen from solid SiO2 in a molten CaCl2 electrolyte at 850 °C. This approach involves a 'contacting electrode', in which a metal wire supplies electrons to a selected region of the insulating SiO2. Bulk reduction of SiO2 is possible by increasing the number of contacting points. The same method was also demonstrated with molten LiCl-KCl-CaCl2 at 500 °C. The novelty and relative simplicity of this method might lead to new processes in silicon semiconductor technology, as well as in high-purity silicon production. The methodology may be applicable to electrochemical processing of a wide variety of insulating materials, provided that the electrolyte dissolves the appropriate constituent ion(s) of the material.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Deal, B.E. & Grove, A.S. General relationship for the thermal oxidation of silicon. J. Appl. Phys. 36, 3770–3778 ( 1965).
Sofield, C.J. & Stoneham, A.M. Oxidation of silicon: the VLSI gate dielectric? Semicond. Sci. Technol. 10, 215–244 ( 1995).
Bauza, D. in Handbook of Surfaces and Interfaces of Materials Vol. 1 (ed. Nalwa, H.S.) 115–216 (Academic, San Diego, USA, 2001).
Zulehner, W., Elvers, B., Hawkins, S., Russey, W. & Schulz, G. (eds) Ullmann's Encyclopedia of Industrial Chemistry 5th edn Vol. A23 721–748 (VCH, Weinheim, 1993).
Srivastava, J.K. & Prasad, M. Electrical conductivity of silicon dioxide thermally grown on silicon, J. Electrochem. Soc. 132, 955–963 ( 1985).
Okabe, T.H., Oishi, T. & Ono, K. Deoxidation of titanium aluminide by Ca-Al alloy under controlled aluminum activity. Metall. Trans. B 23, 583–590 ( 1992).
Okabe, T.H., Deura, T., Oishi, T., Ono, K. & Sadoway, D.R. Thermodynamic properties of oxygen in yttrium-oxygen solid solutions. J. Alloy Comp. 237, 841–847 ( 1996).
Tada, M. & Ito, Y. Passive oxide film growth and pitting corrosion of nickel in LiCl-KCl eutectic melt containing oxide ion. Mater. Sci. Forum 185-188, 917–924 ( 1995).
Chen, G.Z., Fray, D.J. & Farthing, T.W. Direct electrochemical reduction of titanium dioxide to titanium in molten calcium chloride. Nature 407, 361–364 ( 2000).
Chen, G.Z. & Fray, D.J. Cathodic refining in molten salts: Removal of oxygen, sulfur and selenium from static and flowing molten copper. J. Appl. Electrochem. 31, 155–164 ( 2001).
Chen, G.Z., Fray, D.J. & Farthing, T.W. Cathodic deoxygenation of alpha case on titanium and alloys in molten calcium chloride. Metall. Mater. Trans. B 32, 1041–1052 ( 2001).
Massalski, T.B., Okamoto, H., Subramanian, P.R. & Kacprzak, L. (eds) Binary Alloy Phase Diagrams 2nd edn Vol. 3 2915–2918 (ASM International, Materials Park, Ohio, USA, 1990).
Chen, G.Z. & Fray, D.J. in Proc. 6th Int. Symp. Molten Salt Chem. Technol. (eds Nianyi, C & Zhiyu, Q) 79–85 (Shanghai Univ. Press, Shanghai, China, 2001).
Chen, G.Z. & Fray, D.J. in Proc. 4th Int. Conf. Mater. Eng. Res. 1–6 (ICMR, Akita, Japan, 2001).
Kubaschewski, O., Alcock, C.B. & Spencer, P.J. Materials Thermochemistry 6th edn (Pergamon, Oxford, UK, 1993).
Massalski, T.B., Okamoto, H., Subramanian, P.R. & Kacprzak, L. (eds) Binary Alloy Phase Diagrams 2nd edn Vol. 1, 951–954 (ASM International, Materials Park, Ohio, USA, 1990).
Norton, F.J. Permeation of gaseous oxygen through vitreous silica. Nature 191, 701 ( 1961).
Burton, L.C. & Madjid, A.H. Coulomb screening in intrinsic medium-gap semiconductors and the electrical conductivity of silicon at elevated temperatures. Phys. Rev. 185, 1127–1132 ( 1969).
Acknowledgements
We are grateful to L. O. Jerdal for discussions. This study was supported by Yazaki Memorial Foundation for Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Nohira, T., Yasuda, K. & Ito, Y. Pinpoint and bulk electrochemical reduction of insulating silicon dioxide to silicon. Nature Mater 2, 397–401 (2003). https://doi.org/10.1038/nmat900
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat900
This article is cited by
-
Dissolution of Calcium Silicates in Molten CaCl2
Metallurgical and Materials Transactions B (2023)
-
Oxygen evolution behavior of La1−xSrxFeO3−δ electrodes in LiCl–KCl melt
Journal of Applied Electrochemistry (2023)
-
An Electrolysis-Distillation Approach for Producing Potassium Metal
Metallurgical and Materials Transactions B (2023)
-
Boron Removal From Molten Metallurgical Grade Silicon via Flux-Powder Injection
Metallurgical and Materials Transactions B (2023)
-
Electrochemically recycling degraded superalloy and valorizing CO2 in the affordable borate-modified molten electrolyte
Tungsten (2023)