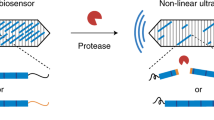
Abstract
Ultrasound is among the most widely used non-invasive imaging modalities in biomedicine1, but plays a surprisingly small role in molecular imaging due to a lack of suitable molecular reporters on the nanoscale. Here, we introduce a new class of reporters for ultrasound based on genetically encoded gas nanostructures from microorganisms, including bacteria and archaea. Gas vesicles are gas-filled protein-shelled compartments with typical widths of 45–250 nm and lengths of 100–600 nm that exclude water and are permeable to gas2,3. We show that gas vesicles produce stable ultrasound contrast that is readily detected in vitro and in vivo, that their genetically encoded physical properties enable multiple modes of imaging, and that contrast enhancement through aggregation permits their use as molecular biosensors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Smith-Bindman, R. et al. Use of diagnostic imaging studies and associated radiation exposure for patients enrolled in large integrated health care systems, 1996–2010. J. Am. Med. Assoc. 307, 2400–2409 (2012).
Walsby, A. E. Gas vesicles. Microbiol. Rev. 58, 94–144 (1994).
Pfeifer, F. Distribution, formation and regulation of gas vesicles. Nature Rev. Microbiol. 10, 705–715 (2012).
Gramiak, R., Shah, P. M. & Kramer, D. H. Ultrasound cardiography—contrast studies in anatomy and function. Radiology 92, 939–948 (1969).
Ferrara, K., Pollard, R. & Borden, M. Ultrasound microbubble contrast agents: fundamentals and application to gene and drug delivery. Annu. Rev. Biomed. Eng. 9, 415–447 (2007).
Kaufmann, B. A. & Lindner, J. R. Molecular imaging with targeted contrast ultrasound. Curr. Opin. Biotechnol. 18, 11–16 (2007).
Cosgrove, D. & Harvey, C. Clinical uses of microbubbles in diagnosis and treatment. Med. Biol. Eng. Comput. 47, 813–826 (2009).
Cui, W. et al. Neural progenitor cells labeling with microbubble contrast agent for ultrasound imaging in vivo. Biomaterials 34, 4926–4935 (2013).
Kircher, M. F., Gambhir, S. S. & Grimm, J. Noninvasive cell-tracking methods. Nature Rev. Clin. Oncol. 8, 677–688 (2011).
Liu, J. et al. Nanoparticles as image enhancing agents for ultrasonography. Phys. Med. Biol. 51, 2179–2189 (2006).
Lanza, G. M. et al. A novel site-targeted ultrasonic contrast agent with broad biomedical application. Circulation 94, 3334–3340 (1996).
Martinez, H. P. et al. Hard shell gas-filled contrast enhancement particles for colour Doppler ultrasound imaging of tumors. Medchemcomm 1, 266–270 (2010).
Kripfgans, O. D., Fowlkes, J. B., Miller, D. L., Eldevik, O. P. & Carson, P. L. Acoustic droplet vaporization for therapeutic and diagnostic applications. Ultrasound Med. Biol. 26, 1177–1189 (2000).
James, M. L. & Gambhir, S. S. A molecular imaging primer: modalities, imaging agents, and applications. Physiol. Rev. 92, 897–965 (2012).
Klebahn, H. Gasvakuolen, ein Bestandteil der Zellen der wasserblutenbildenden Phycochromaceen. Flora (Jena) 80, 241–282 (1895).
Walsby, A. E. Pressure relationships of gas vacuoles. Proc. R. Soc. B 178, 301–326 (1971).
Burns, P. N. Harmonic imaging with ultrasound contrast agents. Clin. Radiol. 51, 50–55 (1996).
De Jong, N., Bouakaz, A. & Frinking, P. Basic acoustic properties of microbubbles. Echocardiography 19, 229–240 (2002).
Hauff, P., Reinhardt, M. & Foster, S. in Molecular Imaging I (eds Semmler, W. & Schwaiger, M.) 223–245 (Springer, 2008).
Perez, J. M., Josephson, L., O'Loughlin, T., Hogemann, D. & Weissleder, R. Magnetic relaxation switches capable of sensing molecular interactions. Nature Biotechnol. 20, 816–820 (2002).
Oliver, R. L. & Walsby, A. E. Direct evidence for the role of light-mediated gas vesicle collapse in the buoyancy regulation of Anabaena flos-aquae (cyanobacteria). Limnol. Oceanogr. 29, 879–886 (1984).
Kashyap, S., Sundararajan, A. & Ju, L. K. Flotation characteristics of cyanobacterium Anabaena flos-aquae for gas vesicle production. Biotechnol. Bioeng. 60, 636–641 (1998).
Geier, M. R., Trigg, M. E. & Merril, C. R. Fate of bacteriophage lambda in non-immune germ-free mice. Nature 246, 221–222 (1973).
Foster, F. S., Pavlin, C. J., Harasiewicz, K. A., Christopher, D. A. & Turnbull, D. H. Advances in ultrasound biomicroscopy. Ultrasound Med. Biol. 26, 1–27 (2000).
Frinking, P. J. & de Jong, N. Acoustic modeling of shell-encapsulated gas bubbles. Ultrasound Med. Biol. 24, 523–533 (1998).
Hoff, L., Sontum, P. C. & Hovem, J. M. Oscillations of polymeric microbubbles: effect of the encapsulating shell. J. Acoust. Soc. Am. 107, 2272–2280 (2000).
Medwin, H. Counting bubbles acoustically: a review. Ultrasonics 15, 7–13 (1977).
Yuan, F. et al. Vascular permeability in a human tumor xenograft: molecular size dependence and cutoff size. Cancer Res. 55, 3752–3756 (1995).
Jokerst, J. V., Lobovkina, T., Zare, R. N. & Gambhir, S. S. Nanoparticle PEGylation for imaging and therapy. Nanomedicine 6, 715–728 (2011).
Li, N. & Cannon, M. C. Gas vesicle genes identified in Bacillus megaterium and functional expression in Escherichia coli. J. Bacteriol. 180, 2450–2458 (1998).
Jost, M., Jones, D. D. & Weathers, P. J. Counting of gas vacuoles by electron microscopy in lysates and purified fractions of Microcystis aeruginosa. Protoplasma 73, 329–335 (1971).
Walsby, A. E. & Armstrong, R. E. Average thickness of the gas vesicle wall in Anabaena flos-aquae. J. Mol. Biol. 129, 279–285 (1979).
Yao, A. I. & Facciotti, M. T. Regulatory multidimensionality of gas vesicle biogenesis in Halobacterium salinarum NRC-1. Archaea 2011, 716456 (2011).
Acknowledgements
The authors thank P. Lum for ultrasound equipment and advice, R. Zalpuri and K. McDonald for assistance with electron microscopy, K-K. Park and P. Khuri-Yakub for assistance with hydrophone measurements, E. Chérin for input on in vivo experiments and the manuscript, and A. Bar-Zion for assistance with data analysis. M.G.S. acknowledges funding from the Miller Research Institute and the Burroughs Wellcome Career Award at the Scientific Interface. Other funding was provided by California Institute for Regenerative Medicine grant RT2-02022 (D.V.S.), National Institutes of Health grant R01EB013689 (S.M.C), the Canadian Institutes of Health Research (F.S.F.) and the Terry Fox Foundation (F.S.F.).
Author information
Authors and Affiliations
Contributions
M.G.S. conceived and directed the study, planned the experiments, prepared the specimens, collected, analysed and interpreted the data, and wrote the manuscript, with input from all other authors. P.W.G. designed and constructed the imaging instrument and accompanying signal processing software, and assisted with initial experiments. A.N. designed, constructed and optimized the imaging instrument and accompanying signal processing software. F.S.F. and M.Y. designed, performed and analysed the data from in vivo experiments. All authors provided input on the study and experimental design, data analysis, data interpretation and the manuscript.
Corresponding author
Ethics declarations
Competing interests
F. Stuart Foster is a consultant to VisualSonics.
Supplementary information
Supplementary information
Supplementary Information (PDF 1058 kb)
Supplementary Movie 1
Supplementary Movie 1 (MOV 10340 kb)
Rights and permissions
About this article
Cite this article
Shapiro, M., Goodwill, P., Neogy, A. et al. Biogenic gas nanostructures as ultrasonic molecular reporters. Nature Nanotech 9, 311–316 (2014). https://doi.org/10.1038/nnano.2014.32
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2014.32
This article is cited by
-
Magneto-acoustic protein nanostructures for non-invasive imaging of tissue mechanics in vivo
Nature Materials (2024)
-
Phase transition of GvpU regulates gas vesicle clustering in bacteria
Nature Microbiology (2024)
-
Wireless agents for brain recording and stimulation modalities
Bioelectronic Medicine (2023)
-
Genomically mined acoustic reporter genes for real-time in vivo monitoring of tumors and tumor-homing bacteria
Nature Biotechnology (2023)
-
Application of Ultrasound to Enhancing Stem Cells Associated Therapies
Stem Cell Reviews and Reports (2023)