Abstract
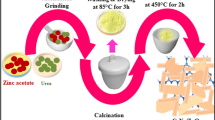
ZnO and 0, 5, and 10 mol % Fe-doped ZnO–TiO2 nanopowders were synthesized by the sol–gel Pechini method. The successful synthesis of coupled ZnO–TiO2 nanopowders was evident by XRD. Scanning electron microscopy (SEM) revealed that the Fe ions were well incorporated into the ZnO–TiO2 crystal lattice. The photocatalytic degradation of anionic surfactant (linear alkylbenzene sulfonate (LABS), was investigated in aqueous solution using ZnO and Fe-doped ZnO–TiO2 nanoparticles. The degradation was studied under different conditions such as the Fe3+ concentration, amount of photocatalyst, irradiation time, pH, initial concentration and presence of electron acceptor. The results showed that photocatalytic degradation of LABS was strongly influenced by these parameters. The best conditions for the photocatalytic degradation of LABS were obtained. It is found that under UV light irradiation, Fe-doping of ZnO–TiO2 increases the efficiency of its photocatalytic activity in degradation of LABS than pure ZnO and ZnO–TiO2.
Similar content being viewed by others
References
S. H. Chiu and G. C. A. Huang, J. Non-Cryst. Solids 358, 2453 (2012).
R. Rajalakshmi and S. Angappane, Mater. Sci. Eng. B 178, 1068 (2013).
P. Hua, N. Hana, D. C. Zhanga, J. Hob, and Y. Chen, Sens. Actuators B 169, 74 (2012).
N. C. Harihara, Appl. Catal. A 30, 455 (2006).
M. A. Behnajady, N. Modirshahla, and R. Hamzayi, J. Hazard Mater. 133, 226 (2006).
R. Y. Hong, T. T. Pan, J. Z. Qian, and H. Z. Li, Chem. Eng. J. 119, 71 (2006).
H. Gerisher and A. Heller, J. Electrochem. Soc. 139, 113 (1992).
U. G. Akpan and B. H. Hameed, Appl. Catal. A: Gen. 375, 1 (2010).
M. L. Zhang, T. C. An, X. H. Hu, C. Wang, G. Y. Sheng, and J. M. Fu, Appl. Catal. A: Gen. 260, 215 (2004).
J. Wang, J. Wan, and K. Chen, Mater. Lett. 64, 2373 (2010).
X. H. Wu, Z. H. Jiang, H. L. Liu, X. D. Li, and X. J. Hu, Mater. Chem. Phys. 80, 39 (2003).
D. E. Gu, B. C. Yang, and Y. D. Hu, Catal. Commun. 9, 1472 (2008).
Y. Li and J. P. Demopoulos, Hydrometallurgy 90, 26 (2008).
C. Shifu and C. Gengyu, Surf. Coat. Technol. 200, 3637 (2006).
M. I. B. Bernardia, L. E. Soledadea, I. A. Santosb, E. R. Leitea, E. Longoa, and J. A. Varelac, Thin Solid Films 405, 228 (2002).
H. Liu, J. Yang, Y. Zhang, L. Yang, M. Wei, and X. Ding, J Phys.: Condens. Matter 21, 145803 (2009).
B. D. Cullity and S. R. Stock, Elements of X-ray Diffraction, 3rd ed. (Prentice-Hall, New Jersey, 2001).
L. Q. Jing, Z. L. Xu, X. J. Sun, J. Shang, and W. M. Cai, Appl. Surf. Sci. 180, 308 (2001).
C. Lee and J. Yoon, J. Photobiol. A: Chem. 165, 35 (2004).
I. K. Konstantinous and T. A. Alabanis, Appl. Catal. B: Environ. 49, 1 (2009).
J. Gimenz, D. Curco, and P. Marco, Water Sci. Technol. 35, 207 (1997).
C. J. da Silva and J. L. Faria, J. Photochem. Photobiol. A: Chem. 155, 133 (2003).
J. M. Hermann, Catal. Today 53, 115 (1999).
B. Rohe, W. S. Veeman, and M. Tausch, Nano Technol. 17, 277 (2006).
U. G. Akpan and B. H. Hameed, J. Hazard. Mater. 170, 520 (2009).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Giahi, M., Saadat Niavol, S., Taghavi, H. et al. Synthesis and characterization of ZnO–TiO2 nanopowders doped with fe via sol–gel method and their application in photocatalytic degradation of anionic surfactant. Russ. J. Phys. Chem. 89, 2432–2437 (2015). https://doi.org/10.1134/S0036024415130154
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024415130154