Abstract
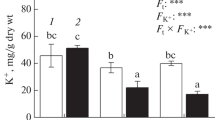
The ameliorative effect of salicilic (SA) and silicon (Si) on photosystem II (PSII) activity of mung bean plant under salt stress (control, 3, 6, and 9 dS/m) was studied by chlorophyll a fluorescence measurement. Results showed that Na+ accumulation decreased, but K+ accumulation increased in mung bean (Vigna radiata (L.) Wilczek) leaves by application of Si and especially SA, leading to improvement in PSII activity under salinity. Initial fluorescence (F 0) and energy necessary for the closure of all reaction centres (S m) were increased with increasing salt levels. Exogenous foliar application of SA and Si decreased initial fluorescence (F 0) and increased photosynthesis relative vitality (PI). Maximum fluorescence (F m), variable fluorescence (F v), the activity of the water-splitting complex on the donor side of the PSII (proportional to F v/F 0) and the average redox state of QA in the time span from 0 to T fm (S m/T fm) were also enhanced by foliar application of SA. Exogenous application of SA improved maximum quantum efficiency of PSII (F v/F m) and performance index (PI) under both saline and non-saline conditions. Increasing in the size of the plastoquinone pool (Area) was much greater for SA than that for Si treated plants. It was concluded that foliar application of Si and SA in particular could play a key role in salt stress tolerance of mung bean plants.
Similar content being viewed by others
Abbreviations
- Area :
-
the area above the fluorescence induction curve between F 0 and F m
- F 0 :
-
initial fluorescence
- F m :
-
maximum fluorescence
- F v :
-
variable fluorescence
- PI :
-
performance index
- PSII:
-
photosystem II
- SA:
-
salicylic acid
- Si:
-
silicon
- S m :
-
energy necessary for the closure of all reaction centres
- T fm :
-
the time span from 0 to F m
References
Lazár, D. and Schansker, G., Models of chlorophyll a fluorescence transients, in Photosynthesis in Silico: Understanding Complexity from Molecules to Ecosystems, vol. 29, Advances in Photosynthesis and Respiration, Laisk, A., Nedbal, L., and Govindjee, Eds., Dordrecht: Springer-Verlag, 2009, pp. 85–123.
van Gorkom, H.J., Fluorescence measurements in the study of photosystem II electron transport, in Light Emission by Plants and Bacteria, Govindjee, Amesz, J., and Fork, D.C., Eds., Ontario: Academic 1986, pp. 267–289.
Schreiber, U., Endo, T., Mi, H., and Asada, K., Quenching analysis of chlorophyll fluorescence by saturation pulse method: particular aspects relating to the study of eukaryotic algae and cyanobacteria, Plant Cell Physiol., 1995, vol. 36, pp. 873–882.
Genty, B., Briantais, J., and Baker, N.R., The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence, Biochim. Biophys. Acta, 1989, vol. 990, pp. 87–92.
Li, R., Guo, P., Baum, M., Grande, S., and Ceccarelli, S., Evaluation of chlorophyll content and fluorescence parameters as indicators of drought tolerance in barley, Agric. Sci. China, 2006, vol. 5, pp. 751–757.
Mehta, P., Jajoo, A., Mathur, S., and Bharti, S., Chlorophyll a fluorescence study reveling effects of high salt stress on photosystem II in wheat leaves, Plant Physiol. Biochem., 2010, vol. 48, pp. 16–20.
Lu, C. and Vonshak, A., Effects of salinity stress on photosystem II function in cyanobacterial Spirulina platensis cells, Physiol. Plant., 2002, vol. 114, pp. 405–413.
Kalaji, M.H. and Guo, P., Chlorophyll fluorescence, a useful tool in barley plant breeding programs, in Photochemistry Research Progress, Sanchez, A. and Gutierrez, S.J., Eds., New York: Nova Publ., 2008, pp. 439–463.
Hasegawa, P.M., Bressan, R.A., Zhu, J.K., and Bohnert, H.J., Plant cellular and molecular responses to high salinity, Plant Mol. Biol., 2000, vol. 51, pp. 463–499.
Senaratna, T., Touchell, D., Bunn, T., and Dixon, K., Acetyl salicylic acid (aspirin) and salicylic acid induce multiple stress tolerance in bean and tomato plants, Plant Growth Regul., 2000, vol. 30, pp. 157–161.
Balakhnina, T. and Borkowska, A., Effects of silicon on plant resistance to environmental stresses: review, Int. Agrophys., 2013, vol. 27, pp. 225–232.
Anwar, F., Latif, S., Przybylski, R., Sultana, B., and Ashraf, M., Chemical composition and antioxidant activity of seeds of different cultivars of mung bean, J. Food Sci., 2007, vol. 72, pp. 503–510.
Munns, R., James, A.J., and Lauchli, A., Approaches to increasing the salt tolerance of wheat and other cereals, J. Exp. Bot., 2006, vol. 57, pp. 1025–1043.
Munns, R. and Tester, M., Mechanism of salinity tolerance, Annu. Rev. Plant Biol., 2008, vol. 59, pp. 651–681.
Semenova, G., Fomina, I., and Ivanov, A., Combined effect of water deficit and salt stress on the structure of mesophyll cells in wheat seedlings, Cell Biol., 2014, vol. 3, pp. 14–24.
Greenway, H. and Munns, R., Mechanisms of salt tolerance in non-halophytes, Annu. Rev. Plant Physiol., 1980, vol. 31, pp. 149–190.
Nishiyama, Y., Los, D.A., and Murata, N., PsbU, a protein associated with photosystem II, is required for the acquisition of cellular thermo tolerance in Synechococcus species PCC 7002, Plant Physiol., 1999, vol. 120, pp. 301–308.
Evans, H.J. and Sorger, G.J., Role of mineral elements with emphasis on univalent cations, Annu. Rev. Plant Physiol., 1966, vol. 17, pp. 47–76.
Yusuf, M., Hasan, S.A., Ali, B., Hayat, S., Fariduddin, Q., and Ahmad, A., Effect of salicylic acid on salinity induced changes in Brassica juncea, J. Integr. Plant Biol., 2007, vol. 50, pp. 1096–1102.
Krause, G.H. and Weis, E., Chlorophyll fluorescence as a tool in plant physiology. II. Interpretation of fluorescence signals, Photosynth. Res., 1984, vol. 5, pp. 139–157.
Havaux, M., Rapid photosynthetic adaptation to heat stress triggered in potato leaves by moderately elevated temperatures, Plant Cell Environ., 1993, vol. 16, pp. 461–467.
Singh-Tomar, R., Mathur, S., Allakhverdiev, S.I., and Jajoo, A., Changes in PS II heterogeneity in response to osmotic and ionic stress in wheat leaves (Triticum aestivum), J. Bioenerg. Biomembr., 2012, vol. 44, pp. 411–419.
Pinior, A., Grunewaldt-Stöcker, G., Alten, H., and Strasser, R.J., Mycorrhizal impact on drought stress tolerance of rose plants probed by chlorophyll a fluorescence, proline content and visual scoring, Mycorrhiza, 2005, vol. 15, pp. 596–605.
Andréasson, L.E., Vass, I., and Styring, S., Ca2+ depletion modifies the electron transfer on the both donor and acceptor sides in photosystem II from spinach, Biochim. Biophys. Acta, 1995, vol. 1230, pp. 155–164.
Enami, I., Kitamura, M., Tomo, T., Isokawa, Y., Ohta, H., and Katch, S., Is the primary cause of thermal inactivation of oxygen evolution in spinach PSII membranes release of the extrinsic 33 kDa protein or of Mn? Biochim. Biophys. Acta, 1994, vol. 1186, pp. 52–58.
Hermans, C., Smeyers, M., Rodriguez, R.M., Eyletters, M., Strasser, R.J., and Delhaye, J.P., Quality assessment of urban trees: a comparative study of physiological characterization, airborne imaging and on site fluorescence monitoring by the O-J-I-P-test, J. Plant Physiol., 2003, vol. 160, pp. 81–90.
Roháček, K., Chlorophyll fluorescence parameters: the definitions, photosynthetic meaning, and mutual relationship, Photosynthetica, 2002, vol. 40, pp. 13–29.
Netondo, G.W., Onyango, J.C., and Beck, E., Sorghum and salinity. II. Gas exchange and chlorophyll fluorescence of sorghum under salt stress, Crop Sci., 2004, vol. 44, pp. 806–811.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Ghassemi-Golezani, K., Lotfi, R. The impact of salicylic acid and silicon on chlorophyll a fluorescence in mung bean under salt stress. Russ J Plant Physiol 62, 611–616 (2015). https://doi.org/10.1134/S1021443715040081
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443715040081