Abstract
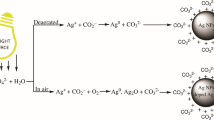
Silver nanoparticles are prepared by reducing Ag+ ions with sodium borohydride in aqueous solutions containing carbonate ions (5 × 10−5−1 × 10−2 mol l−1). It is established that carbonate ions represent an efficient stabilizer that provides nanoparticles with electrostatic protection via the formation of an electrical double layer. The maximum stability of a silver dispersion is observed at a carbonate ion concentration of 1 × 10−3 mol l−1. The average size of silver nanoparticles is 10.0 ± 2.5 nm. The formation kinetics of silver nanoparticles is described by an equation for a first-order reaction with a rate constant of 2.3 × 10−3 s−1 (±20%).
Similar content being viewed by others
References
Gubin, S.P., Yurkov, G.Yu., and Kataeva, N.A., Nanochastitsy blagorodnykh metallov (Noble Metal Nanoparticles), Moscow: OOO Azbuka, 2000.
Summ, B.D. and Ivanova, N.I., Usp. Khim., 2000, vol. 69, p. 995.
Carlotti, M.E., Morel, S., Cavalli, R., and Minero, C., J. Dispersion Sci. Technol., 1993, vol. 14, p. 35.
Van der Leeden, M.C. and Van Rosmalen, G.M., J. Colloid Interface Sci., 1995, vol. 171, p. 142.
Fendler, J.H., Chem. Rev., 1987, vol. 87, p. 877.
Sergeev, B.M., Kiryukhin, M.V., Bakhov, F.N., and Sergeev, V.G., Vestn. Mosk. Univ., Ser. 2: Khim., 2001, vol. 42, p. 308.
Peyre, V., Spalla, O., Beloni, L., and Nabavi, M., J. Colloid Interface Sci., 1997, vol. 187, p. 184.
Spalla, O. and Kekicheff, P., J. Colloid Interface Sci., 1997, vol. 192, p. 43.
Cason, J.P., Khambaswadkar, K., and Roberts, C.B., Ind. Eng. Chem. Res., 2000, vol. 39, p. 4749.
Maillard, M., Giorgio, S., and Pileni, M.-P., J. Phys. Chem. B, 2003, vol. 107, p. 2466.
Chen, S. and Carroll, D.L., Nano Lett., 2002, vol. 2, p. 1003.
Li, X., Zhang, J., Xu, W., et al., Langmuir, 2003, vol. 19, p. 4285.
He, S., Yao, J., Jiang, P., et al., Langmuir, 2001, vol. 17, p. 1571.
Chen, D.-H. and Huang, Y.-W., J. Colloid Interface Sci., 2002, vol. 255, p. 299.
Waters, C., Mills, A.J., Johnson, K.A., and Schiffrin, D.J., Chem. Commun., 2005, p. 787.
Ershov, B.G., Abkhalimov, E.V., and Sukhov, N.L., Khim. Vys. Energ., 2005, vol. 39, p. 55.
Bohren, C.F. and Huffman, D.R., Absorption and Scattering of Light by Small Particles, New York: Wiley, 1983.
Kerker, M., The Scattering of Light and Other Electromagnetic Radiation, New York: Academic, 1969, p. 38.
Ershov, B.G., Ross. Khim. Zh., 2001, vol. 45, no. 3, p. 20.
Henglein, A., Mulvaney, P., and Linnert, T., J. Chem. Soc., Faraday Discuss., 1991, vol. 92, p. 31.
Ershov, B.G. and Gordeev, A.V., Mendeleev Commun., 2001, no. 4, p. 147.
Ung, T., Giersig, M., Dunstan, D., and Mulvaney, P., Langmuir, 1997, vol. 13, p. 1773.
Chou, K-S. and Lai, Y-S., Mater. Chem. Phys., 2004, vol. 83, p. 82.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.V. Abkhalimov, A.A. Parsaev, B.G. Ershov, 2011, published in Kolloidnyi Zhurnal, 2011, Vol. 73, No. 1, pp. 3–8.
Rights and permissions
About this article
Cite this article
Abkhalimov, E.V., Parsaev, A.A. & Ershov, B.G. Preparation of silver nanoparticles in aqueous solutions in the presence of carbonate ions as stabilizers. Colloid J 73, 1–5 (2011). https://doi.org/10.1134/S1061933X11010029
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X11010029