Abstract
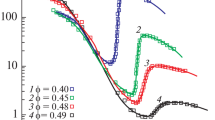
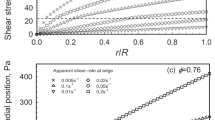
The rheological properties of concentrated suspensions of metal oxides dispersed in transformer oil, which are used as electrorheological fluids, are systematically studied. Colloidal particles have intermediate sizes between nano- and microsized scales. Low-amplitude dynamic measurements show that the storage moduli of the examined suspensions are independent of frequency and these materials should be considered as solidlike elastic media. The storage modulus is proportional to the five-powdered particle volume concentration. At the same time, a transition through an apparent yield stress with a reduction in the viscosity by approximately six orders of magnitude is distinctly seen upon shear deformation. The character of the rheological behavior depends on the regime of suspension deformation. At very low shear rates, a steady flow is possible; however, upon an increase in the rate, an unsteady regime is realized with development of self-oscillations. When constant shear stresses are preset, in some range of stresses, thickening of the medium takes place, which can also be accompanied by self-oscillations. In order to gain insight into the nature of this effect, measurements are performed for samples with different volume/surface ratios, which show that, in some deformation regimes, suspension is separated into layers and slipping occurs along a low-viscosity layer with a thickness of several dozen microns. Direct observations show a distinct structural inhomogeneity of the flow. The separation and motion of layers with different compositions explain the transition to the flow with the lowest apparent Newtonian viscosity. Thus, the deformation of concentrated suspensions is associated with self-oscillations of stresses and slipping along a low-viscosity interlayer.
Similar content being viewed by others
References
Abduragimova, L.A., Rehbinder, P.A., and Serb-Serbina, N.N., Kolloidn. Zh., 1955, vol. 17, p. 184.
Heymann, L., Peukert, S., and Aksel, N., Rheol. Acta, 2002, vol. 41, p. 307.
Moan, M., Aubry, T., and Bossard, F., J. Rheol. (N. Y.), 2003, vol. 47, p. 1493.
Potanin, A., J. Rheol. (N. Y.), 2004, vol. 48, p. 1279.
Dullaert, K. and Mewis, J., J. Rheol. (N. Y.), 2005, vol. 49, p. 1213.
Stokes, J.R., Telford, J.H., and Williamson, A.-M., J. Rheol. (N. Y.), 2005, vol. 49, p. 139.
Coussot, P., Chateau, X., Tocquer, L., et al., J. Rheol. (N. Y.), 2006, vol. 50, p. 975.
Franks, G.V., Zhou, Zh., Duin, N.J., and Boger, D.V., J. Rheol. (N. Y.), 2000, vol. 44, p. 759.
Egres, R.G. and Wagner, N.J., J. Rheol. (N. Y.), 2005, vol. 49, p. 719.
Uhlherr, P.H.T., Guo, J., Tiu, C., et al., J. Non-Newtonian Fluid Mech., 2005, vol. 125, p. 101.
Khan, S.A. and Zoeller, N.J., J. Rheol. (N. Y.), 1993, vol. 37, p. 1225.
Heymann, L., Peukert, S., and Aksel, N., J. Rheol. (N. Y.), 2002, vol. 46, p. 93.
O’Brien, V.T. and Mackley, M.E., J. Rheol. (N. Y.), 2002, vol. 46, no. 3, p. 557.
Lee, Y.S. and Wagner, N.J., Rheol. Acta, 2003, vol. 42, p. 199.
Il’in, S.O., Malkin, A.Ya., Korobko, E.V., et al., J. Eng. Phys. Thermophys., 2011, vol. 84, p. 1016.
Buscall, R., Mills, P.D.A., Goodwin, J.W., and Lawson, D.W., J. Chem. Soc., Faraday Trans., 1988, vol. 84, p. 4249.
Wyss, H.M., Deliormanli, A.M., Tervoort, E., and Gauckler, L.J., AIChE J., 2005, vol. 51, p. 134.
Pavlov, V.P. and Vinogradov, G.V., Kolloidn. Zh., 1966, vol. 28, p. 424.
Shalopalkina, T.G. and Trapeznikov, A.A., Kolloidn. Zh., 1960, vol. 22, p. 735.
Mustafaev, E., Malkin, A.Ya., Plotnikova, E.P., and Vinogradov, G.V., Vysokomol. Soedin., 1964, vol. 6, p. 1515.
Boltenhagen, P., Hu, Y., Matthys, E.F., and Pine, D.J., Europhys. Lett., 1997, vol. 38, p. 389.
Wunenburger, A.S., Colin, A., Leng, J., et al., Phys. Rev. Lett., 2001, vol. 86, p. 1374.
Lootens, D., Damme, H.V., and Hebraud, P., Phys. Rev. Lett., 2003, vol. 90, p. 178301.
Bagusat, F., Bohme, B., Schiller, P., and Mogel, H.-J., Rheol. Acta, 2005, vol. 44, p. 313.
Malkin, A.Ya., Semakov, A.V., and Kulichikhin, V.G., Adv. Colloid Interface Sci., 2010, vol. 157, p. 75.
Bashkirtseva, I.A., Zubarev, A.Yu., Iskakova, L.Yu., and Ryashko, L.B., Kolloidn. Zh., 2010, vol. 72, p. 147.
Subbotin, A.V., Malkin, A.Ya., and Kulichikhin, V.G., Adv. Colloid Interface Sci., 2011, vol. 162, p. 29.
Berret, J.-F., Roux, D.C., and Porte, G., J. Phys. II, 1994, vol. 4, p. 1261.
Lerouge, S. and Berret, J.-F., Adv. Polym. Sci., 2010, vol. 230, p. 1.
Hu, Y.T., Palla, C., and Lips, A., J. Rheol. (N. Y.), 2008, vol. 52, p. 379.
Zhou, L., Vasquez, P.A., Cook, L.P., and McKinley, G.H., J. Rheol. (N. Y.), 2008, vol. 52, p. 591.
Tapadia, P., Ravindranath, S., and Wang, S.-Q., Phys. Rev. Lett., 2006, vol. 96, p. 196001.
Ilyin, S., Roumyantseva, T., Spiridonova, V., et al., Soft Matter, 2011, vol. 7, p. 9090.
Ur’ev, N.B., Colloids Surf. A., 1994, vol. 8, p. 1.
Ur’ev, N.B., Uspekhy Khimii, 2004, vol. 73, p. 39.
Malkin, A.Ya. and Chalykh, A.E., Diffuziya i vyazkost’. Metody izmerenii (Diffusion and Viscosity. Measurement Methods), Moscow: Khimiya, 1979.
Chen, L.B., Zukoski, C.F., Ackerson, B.J., et al., Phys. Rev. Lett., 1992, vol. 69, p. 688.
Vermant, J. and Solomon, M.J., J. Phys.: Condens. Matter, 2005, vol. 17, p. 187.
Miesowicz, M., Nature (London), 1935, vol. 136, p. 261.
Miesowicz, M., Nature (London), 1946, vol. 158, p. 27.
Author information
Authors and Affiliations
Additional information
Original Russian Text © S.O. Ilyin, A.Ya. Malkin, V.G. Kulichikhin, 2012, published in Kolloidnyi Zhurnal, 2012, Vol. 74, No. 4, pp. 492–502.
Rights and permissions
About this article
Cite this article
Ilyin, S.O., Malkin, A.Y. & Kulichikhin, V.G. Rheological peculiarities of concentrated suspensions. Colloid J 74, 472–482 (2012). https://doi.org/10.1134/S1061933X12040072
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061933X12040072