Abstract
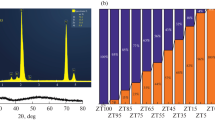
Formation of zirconia nanocrystals in the course of thermal treatment of an X-ray amorphous zirconium oxyhydroxide was studied. It was shown that the formation of tetragonal and monoclinic polymorphs of ZrO2 in the temperature range from 500 to 700°C occurs owing to dehydration and crystallization of amorphous hydroxide. An increase of the temperature up to 800°C and higher activates mass transfer processes and, as a result, activates the nanoparticle growth and increases the fraction of the phase based on monoclinic modification of ZrO2 due to mass transfer from the nanoparticles with the non-equilibrium tetragonal structure. Herewith, formed ZrO2 nanocrystals with monoclinic structure have a broad size distribution of crystallites, and the average crystallite size after thermal treatment at 1200°C for 20 min is about 42 nm.
Similar content being viewed by others
References
Nanomaterialy: svoistva i perspektivnye prilozheniya (Nanomaterials: Properties and Potential Applications), Yaroslavtsev, A.B., Ed., Moscow: Nauchnyi Mir, 2014.
Shukla, S. and Seal, S., Int. Mater. Rev., 2005, vol. 50, no. 1, p. 45. DOI: 10.1179/174328005X14267.
Clament Sagaya Selvama, N., Manikandana, A., John Kennedy, L., and Judith Vijaya, J., J. Colloid Interf. Sci., 2013, vol. 389, no. 1, p. 91. DOI: 10.1016/ j.jcis.2012.09.014.
Demortiere, A., Panissod, P., Pichon, B.P., Pourroy, G., Guillon, D., Donnio, B., and Begin-Colin, S., Nanoscale, 2011, vol. 3, p. 225. DOI: 10.1039/c0nr00521e.
Uchino, K., Sadanaga, E., and Hirose, T., J. Am. Ceram. Soc., 1989, vol. 72, no. 8, p. 1555. DOI: 10.1111/j.1151- 2916.1989. tb07706.x.
Katz, G., J. Am. Ceram. Soc., 1971, vol. 54, no. 10, p. 531. DOI: 10.1111/j.1151-2916.1971.tb12197.x.
Pozhidaeva, O.V., Korytkova, E.N., Drozdova, I.A., and Gusarov, V.V., Russ. J. Gen. Chem., 1999, vol. 69, no. 8, p. 1219. DOI: 10.1134/S107036329908006X.
Pentin, I.V., Oleinikov, N.N., Murav’eva, G.P., Eliseev, A.A., and Tret’yakov, Yu.D., Inorg. Mater., 2002, vol. 38, no. 10, p. 1012. DOI: 10.1023/A:1020521220136.
Dwivedi, R., Maurya, A., Verma, A., Prasad, R., and Bartwal, K.S., J. Alloys Compd., 2011, vol. 509, p. 6848. DOI: 10.1016/j.jallcom.2011.03.138.
Li, F., Li, Y., Song, Z., Ma, F., Xu, K., and Cui, H., J. Eur. Ceram. Soc., 2015, vol. 35, no. 8, p. 2361. DOI: 10.1016/j. jeurceramsoc.2015.02.017.
Belov, G.V., Iorish, V.S., and Yungman, V.S., CALPHAD, 1999, vol. 23, no. 2, p. 173. DOI: 10.1016/ S0364-5916(99)00023-1.
Ruff, O. and Ebert, F., Z. Anorg. Allg. Chem., 1929, vol. 180, no. 1, p. 19. DOI:10.1002/zaac.19291800104.
Gundiah, G., Mkhopadhyay, S., Tomkurkar, U.G., Govindara, A., Maitra, U., and Rao, C.N.R., J. Mater. Chem., 2003, vol. 13, p. 2118. DOI:10.1039/B304007K.
Burda, C., Chen, X., Narayanan, R., and Sayed, M.A., Chem. Rev., 2005, vol. 105, no. 4, p.1025. DOI: 10.1021/cr030063a.
Lin, F.-Q., Dong, W.-S., Liu, C.-L., Liu, Z., and Li, M., J. Colloid Interf. Sci., 2008, vol. 323, no. 2, p. 365. DOI: 10.1016/j.jcis.2008.04.030.
Bugrov, A.N. and Almjasheva, O.V., Nanosystems: Physics, Chemistry, Mathematics, 2013, vol. 4, no. 6, p. 810.
Kumar, S., Bhunia, S., and Ojha, A.K., Physica E, 2015, vol. 66, p. 74. DOI: 10.1016/j.physe.2014.09.007.
Oleinikov, N.N., Pentin, I.V., Murav’eva, G.P., and Ketsko, V.A., Russ. J. Inorg. Chem., 2001, vol. 46, no. 9, p. 1275.
Davis, B.H., Commun. Am. Ceram. Soc., 1984, vol. 67, no. 8, p. 168. DOI: 10.1111/j.1151-2916.1984.tb19185.x.
Srinivasan, R., Davis, B.H., Cavin, O.B., and Hubbard, C.R., J. Am. Ceram. Soc., 1992, vol. 75, no. 5, p. 1217. DOI:10.1111/j.1151-2916.1992.tb05560.x.
Gusarov, V.V. and Suvorov, S.A., Zh. Prikl. Khim., 1987, vol. 60, no. 12, p. 1953.
Almjasheva, O.V. and Gusarov, V.V., Int. J. Alternative Energy Ecol., 2007, no. 113, p. 113.
Almjasheva, O.V., Nanosystems: Physics, Chemistry, Mathematics, 2015, vol. 6, no. 5, p. 697. DOI: 10.17586/2220-8054-2015-6-5-697-703.
Lomanova, N.A. and Gusarov, V.V., Nanosystems: Physics, Chemistry, Mathematics, 2013, vol. 4, no. 5, p. 696.
Lutterotti, L., Nucl. Instrum. Meth. B, 2010, vol. 268, p. 334. DOI: 10.1016/j.nimb.2009.09.053.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.K. Vasilevskaya, O.V. Almjasheva, V.V. Gusarov, 2015, published in Zhurnal Obshchei Khimii, 2015, Vol. 85, No. 12, pp. 1937–1941.
Rights and permissions
About this article
Cite this article
Vasilevskaya, A.K., Almjasheva, O.V. & Gusarov, V.V. Formation of nanocrystals in the ZrO2–H2O system. Russ J Gen Chem 85, 2673–2676 (2015). https://doi.org/10.1134/S1070363215120014
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363215120014