Abstract
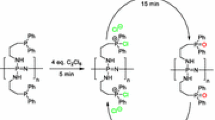
Reaction of hexachlorocyclotriphosphazene and a mixture of cyclic chlorophosphazene [NPCl2] n = 3–8 with an excess of diphenylolpropane under different conditions affords corresponding oligomeric hydroxy-aryloxy phosphazenes, which were characterized by gas chromatography-mass spectrometry, laser mass spectrometry, 31P and 1H NMR spectroscopy. Side reactions was found with participation of decomposition products of diphenylolpropane.
Similar content being viewed by others
References
Kireev, V.V., Chistyakov, E.M., Filatov, S.N. et al., Polymer Sci., Ser. A, 2011, vol. 53, no. 7, pp. 412–419.
Chuev, V.P., Gapochkina, L.L., Kireev, V.V. et al., In-t Stomatologii, 2010, no. 3, pp. 94–96.
RF Patent 2375039, 2008.
Bilichenko, Yu.V., Cand. Sci. (Chem.) Dissertation, Moscow, 2008.
Sirotin, I.S., Bilichenko, Yu.V., Suraeva, O.V. et al., Polymer Sci., Ser. A, 2013, vol. 55, no. 2, pp. 63–68.
Weissberger, A., Proskauer, E.S., Riddick, J.A., and Toops, E.E., Jr., Organic Solvents: Physical Properties and Methods of Purification, New York: Intersci., 1955, 2nd ed.
Verkhovskaya, Z.N., Difenilolpropan (Diphenylolpropane), Moscow: Khimiya, 1971.
Gapochkina, L.L., Cand. Sci. (Chem.) Dissertation, Moscow, 2009.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.S. Sirotin, Yu.V. Bilichenko, K.A. Brigadnov, V.V. Kireev, O.V. Suraeva, R.S. Borisov, 2013, published in Zhurnal Prikladnoi Khimii, 2013, Vol. 86, No. 12, pp. 1956–1965.
Rights and permissions
About this article
Cite this article
Sirotin, I.S., Bilichenko, Y.V., Brigadnov, K.A. et al. Oligomeric hydroxy-aryloxy phosphazene based on cyclic chlorophosphazenes. Russ J Appl Chem 86, 1903–1912 (2013). https://doi.org/10.1134/S1070427213120161
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427213120161