Abstract
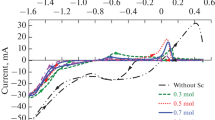
Method of potentiodynamic polarization was used to study the electrodeposition of a copper-tin alloy from an oxalic acid electrolyte with addition of 10−5–10−3 mol dm−3 of trimethyloctylammonium chloride, N-octylpyridinium bromide, and N-benzylpyridinium bromide. Introduction of these compounds changes the rate of the cathodic reduction of tin. It was found that the nature and concentration of the additives introduced into the electrolyte affects the composition, structure, glossiness, and roughness of the Cu-Sn coatings being formed. It was shown that introduction into an electrolyte of trimethylammonium chloride and N-octylpyridinium bromide inhibits the deposition of tin and favors formation of lustrous and semilustrous coatings containing 24.4–35.8 wt % tin. The presence of N-benzylpyridinium bromide in an oxalic acid electrolyte intensifies the cathodic reduction of tin and leads to formation of matte coatings containing up to 42.5 wt % tin.
Similar content being viewed by others
References
Finazzi, G.A., de Oliveria, E.M., and Carlos, I.A., Surf. Coat. Technol., 2004, vol. 187, pp. 377–387.
Walsh, F.C. and Low, C.T.J., Surf. Coat. Technol., 2016, vol. 304, pp. 246–262.
Antikhovich, I.V., Kharitonov, D.S., Chernik, A.A., and Dobryden, I.B., Russ. J. Appl. Chem., 2017, vol. 90, no. 4 pp. 566–573.
Bogomazova, N.V., Antikhovich, I.V., Chernik, A.A., and Zharskii, I.M., Russ. J. Appl. Chem., 2014, vol. 87, no. 9 pp. 1235–1239.
Hovestad, A., Tacken, R.A., and Mannetje, H.H., Phys. Status Solidi, 2008, vol. 5, pp. 3506–3509.
Møller, P., Rasmussen, J.B., Köhler, S., Elplatek, A., and Nielsen, L.P., NASF Surf. Technol., White Papers, 2013, vol. 78, pp. 15–24.
Piccinini, N., Ruggiero, G.N., Baldi, G., and Robotto, A., J. Hazard. Mater., 2000, vol. 71, pp. 395–407.
Bengoa, L.N., Tuckart, W.R., Zabala, N., Prieto, G., and Egli, W.A., Surf. Coat. Technol., 2014, vol. 253, pp. 241–248.
Kasach, A.A., Kurilo, I.I., Kharitonov, D.S., Radchenko, S.L., and Zharskii, I.M., Russ. J. Appl. Chem., 2018, vol. 91, no. 4 pp. 591–596.
Medvedev, G.I. and Makrushin, N.A., Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol., 2009, no. 12, pp. 61–65.
Yakovleva, A.A., Chyong, S.N., Pridatchenko, Yu.V., and Shuvaeva, E.M., Izv. Vyssh. Uchebn. Zaved., Khim. Khim. Tekhnol., 2013, no. 1(4), pp. 106–111.
Acknowledgments
The authors are grateful to V.S. Bezborodov, professor of the department of organic chemistry at the Belarussian State Technological University, for assistance in syntheses of quaternary ammonium salts examined in the study.
Funding
The study was financially supported by the Ministry of Education of the Republic of Belarus (grant “Electrochemical composite coatings with photocatalytic properties, based on tin alloys”).
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © The Author(s), 2019, published in Zhurnal Prikladnoi Khimii, 2019, Vol. 92, No. 6, pp. 793–799.
Conflict of Interest
The authors state that they have no conflict of interest to be disclosed in this communication.
Rights and permissions
About this article
Cite this article
Kasach, A.A., Kharitonov, D.S., Romanovskii, V.I. et al. Electrodeposition of Cu-Sn Alloy from Oxalic Acid Electrolyte in the Presence of Amine-containing Surfactants. Russ J Appl Chem 92, 835–841 (2019). https://doi.org/10.1134/S1070427219060144
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427219060144