Abstract
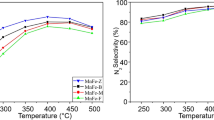
The properties of the Mn–Ce/Beta zeolite catalyst in the selective catalytic reduction (SCR) of NO x have been investigated. The introduction of Ce leads to a marked increase in the NO x conversion at 100–250°C. The data of this study are consistent with the “bifunctional” pathway of SCR suggested for Mn/Beta, which consists of two stages—NO oxidation to NO2 over the oxide component and “fast” SCR over the zeolite. The increased activity of Mn–Ce/Beta at the NO-to-NO2 oxidation stage is due to the formation of MnCeO x mixed oxides enhancing the mobility of lattice oxygen. The determining role is played by the activity of the zeolite component in the “fast” SCR reaction.
Similar content being viewed by others
References
Bosch, H. and Janssen, F., Catal. Today, 1988, vol. 2, p. 369.
Zhu, Z., Liu, Z., Niu, H., and Liu, S., J. Catal., 1999, vol. 187, p. 245.
Parvulescu, V.I., Grange, P., and Delmon, B., Catal. Today, 1998, vol. 46, p. 233.
Carja, G., Kameshima, Y., Okada, K., and Madhusoodana, C.D., Appl. Catal., B, 2007, vol. 73, p. 60.
Yang, S.J., Li, J.H., Wang, C.Z., Chen, J.H., Ma, L., Chang, H.Z., Chen, L., Peng, Y., and Yan, N.Q., Appl. Catal. B, 2012, vol. 117, p. 73.
Qi, G.S., Yang, R.T., and Chang, R., Appl. Catal. B, 2004, vol. 51, p. 93.
Singoredjo, L., Korver, R., Kapteijn, F., and Moulijn, J., Appl. Catal. B, 1992, vol. 1, p. 297.
Kijlstra, W.S., Brands, D.S., Poels, E.K., and Bliek, A., Catal. Today, 1999, vol. 50, p. 133.
Ramis, G., Yi, L., Busca, G., Turco, M., Kotur, E., and Willey, R.J., J. Catal., 1995, vol. 157, p. 523.
Huang, H., Long, R.Q., and Yang, R.T., Appl. Catal. A, 2002, vol. 235, p. 241.
Kumar, M.S., Schwidder, M., Grunert, W., and Brückner, A., J. Catal., 2004, vol. 227, p. 384.
Krivoruchenko, D.S., Kucherov, A.V., Telegina, N.S., Bokarev, D.A., Selvam, P., and Stakheev, A.Yu., Russ. Chem. Bull., 2014, vol. 63, p. 389.
Salazar, M., Becker, R., and Grünert, W., Appl. Catal. B, 2015, vol. 165, p. 316.
Stakheev, A.Yu., Baeva, G.N., Bragina, G.O., Teleguina, N.S., Kustov, A.L., Grill, M., and Thogersen, J.R., Top. Catal., 2013, vol. 56, p. 427.
Zhang, L., Pierce, J., Leung, V.L., Wang, D., and Epling, W.S., J. Phys. Chem. C, 2013, vol. 117, p. 8282.
Qi, G.S. and Yang, R.T., J. Catal., 2003, vol. 217, p. 434.
Qi, G.S. and Yang, R.T., J. Phys. Chem. B, 2004, vol. 108, p. 15738.
Cao, F., Su, S., Xiang, J., Wang, P., Hu, S., Sun, L., and Zhang, A., Fuel, 2015, vol. 139, p. 232.
Hong, W.J., Iwamoto, S., and Inoue, M., Catal. Today, 2011, vol. 164, p. 489.
Liu, Y., Gu, T.T., Weng, X.L., Wang, Y., Wu, Z.B., and Wang, H.Q., J. Phys. Chem. C, 2012, vol. 116, p. 16582.
Wu, Z.B., Jiang, B.Q., Liu, Y., Zhao, W.R., and Guan, B.H., J. Hazard. Mater., 2007, vol. 145, p. 488.
Machida, M., Uto, M., Kurogi, D., and Kijima, T., Chem. Mater., 2000, vol. 12, p. 3158.
Mytareva, A.I., Baeva, G.N., Bragina, G.O., Selvam, P., Bokarev, D.A., and Stakheev, A.Yu., Mendeleev Commun., 2014, vol. 24, p. 311.
Zhang, R., Zhang, B., Shi, Z., Liu, N., and Chen, B., J. Mol. Catal. A: Chem., 2015, vol. 398, p. 223.
You, Z., Balint, I., and Aika, K., Appl. Catal., B, 2004, vol. 53, p. 233.
Nie, J., Wu, X., Ma, Z., Xu, T., Si, Z., Chen, L., and Weng, D., Chin. J. Catal., 2014, vol. 35, p. 1281.
Sun, M., Lan, B., Yun, L., Ye, F., Song, W., He, J., Diao, G., and Zheng, Y., Mater. Lett., 2012, vol. 86, p. 18.
Lou, X., Liu, P., Li, J., and He, K., Appl. Surf. Sci., 2014, vol. 307, p. 382.
Liu, J., Yu, C.Y., Zhao, P.Q., and Chen, G.X., Appl. Surf. Sci., 2012, vol. 258, p. 9096.
Tang, X., Li, Y., Huang, X., Xu, Y., Zhu, H., Wang, J., and Shen, W., Appl. Catal., B, 2006, vol. 62, p. 265.
Machida, M., Catal. Surv. Jpn., 2002, vol. 5, p. 91.
Shi, L., Chu, W., Qu, F., and Luo, S., Catal. Lett., 2007, vol. 113, p. 59.
Wu, Z., Jin, R., Liu, Y., and Wang, H., Catal. Commun., 2008, vol. 9, p. 2217.
Xingyi, W., Qian, K., and Dao, L., Appl. Catal., B, 2009, vol. 86, p. 166.
Qi, G., Yang, R.T., and Chang, R., Catal. Lett., 2003, vol. 87, p. 67.
Tang, X., Hao, J., Yi, H., and Li, J., Catal. Today, 2007, vol. 126, p. 406.
Delimaris, D. and Ioannides, T., Appl. Catal., B, 2008, vol. 84, p. 303.
Li, W.B., Wang, J.X., and Gong, H., Catal. Today, 2009, vol. 148, p. 81.
Saqer, S.M., Kondarides, D.I., and Verykios, X.E., Appl. Catal., B, 2011, vol. 103, p. 275.
Liu, X., Lu, J., Qian, K., Huang, W., and Luo, M., J. Rare Earths, 2009, vol. 27, p. 418.
Zou, Z.Q., Meng, M., and Zha, Y.Q., J. Phys. Chem. C, 2009, vol. 114, p. 468.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © D.S. Krivoruchenko, N.S. Telegina, D.A. Bokarev, A.Yu. Stakheev, 2015, published in Kinetika i Kataliz, 2015, Vol. 56, No. 6, pp. 729–734.
Rights and permissions
About this article
Cite this article
Krivoruchenko, D.S., Telegina, N.S., Bokarev, D.A. et al. Mn–Ce/beta “bifunctional” catalyst for the selective catalytic reduction of nitrogen oxides with ammonia. Kinet Catal 56, 741–746 (2015). https://doi.org/10.1134/S0023158415060051
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158415060051