Abstract
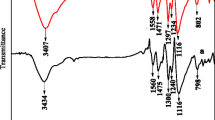
A facile approach has been established to prepare PPy via in situ polymerization with different metal sulfate as dopants. The morphology and structure of PPy and doped PPy were characterized by scanning electron microscopy (SEM) and fourier transform infrared (FT-IR). It was found that doped PPy has different morphology and a slight structure change. The electrochemical performance of the samples has been illustrated by cyclic voltammetry (CV), galvanostatic charge–discharge (GCD) and AC impedance measurements. Compared with the PPy, the specific capacitance of PPy/Cu2+ has been improved to 224 F g–1 at the current density of 0.6 A g–1. Also, the relationship between electrochemical properties of doped PPy and various parameters of metal ions has been investigated.
Similar content being viewed by others
References
Cho, S.I. and Sang, B.L., Accounts Chem. Res., 2008, vol. 41, pp. 699–707.
Vernitskaya, T.V. and Efimov, O.N., Cheminform, 1997, p. 28.
Syed, A.A. and Dinesan, M.K., Talanta, 1991, vol. 38, pp. 815–837.
Mccullough, R.D., Adv. Mater., 1998, vol. 10, pp. 93–116.
Tamm, J., Raudsepp, T., Marandi, M., and Tamm, T., Synthetic Metals, 2007, vol. 157, pp. 66–73.
Linv, S., New Chem. Mater., 2011, vol. 39, pp. 31–34.
Zhang, A.Q., Wang, L.Z., Zhang, Y., and Shen, F.B., Battery Bimonthly, 2010, vol. 40, pp. 10–12.
Hong-Yu, M.I., Zhang, X.G., Xin-Mei, L.V., Luo, J.M., and Huang, J.S., Polym. Mater. Sci. Eng., 2008, vol. 24, pp. 155–158.
Liang, L.I., Zhu, H.B., Dan, Y.U., and Chen, X., J. Wuhan Inst. Technol., 2013, vol. 35, no. 5, pp. 43–46.
Dimitriev, O.P., Polym. Bull., 2003, vol. 50, pp. 83–90.
Wang, Y., Yang, C., and Liu, P., Chem. Eng. J., 2011, vol. 172, pp. 1137–1144.
Jayamurgan, P., Ponnuswamy, V., Ashokan, S., and Mahalingam, T., Iran. Polym. J., 2013, vol. 22, pp. 219–225.
Liang, L.I., Lan-Yan, L.I., and Wang, P., J. Wuhan Inst. Technol., 2013, vol. 35, no. 3, pp. 43–47.
Yang, C. and Liu, P., Synthetic Metals, 2010, vol. 160, pp. 768–773.
Sahoo, S., Dhibar, S., Hatui, G., Bhattacharya, P., and Das, C.K., Polymer, 2013, vol. 54, pp. 1033–1042.
Xu, H., Zhang, J., Chen, Y., Lu, H., and Zhuang, J., J. Solid State Electrochem., 2014, vol. 18, pp. 813–819.
Zhang, D., Zhang, X., Chen, Y., Yu, P., Wang, C., and Ma, Y., J. Power Sources, 2011, vol. 196, pp. 5990–5996.
Dubal, D., Patil, S., Jagadale, A., and Lokhande, C., J. Alloys Compd., 2011, vol. 509, pp. 8183–8188.
Brousse, T., Toupin, M., Dugas, R., Athou, L., Crosnier, O., and Bélanger, D., J. Electrochem. Soc., 2006, vol. 153, pp. A2171–A2180.
Dubal, D.P., Lee, S.H., Kim, J.G., Kim, W.B., and Lokhande, C.D., J. Mater. Chem., 2012, vol. 22, pp. 3044–3052.
Xu, M., Kong, L., Zhou, W., and Li, H., J. Phys. Chem. C, 2007, vol. 111, pp. 19141–19147.
Xu, H., Wu, J., Li, C., Zhang, J., and Liu, J., Electrochim. Acta, 2015, vol. 165, pp. 14–21.
Xu, H., Li, J., Peng, Z., Zhuang, J., and Zhang, J., Electrochim. Acta, 2013, vol. 90, pp. 393–399.
Sheng, L.I., Qiu, Y.B., and Guo, X.P., Acta Phys.-Chim. Sinica, 2010, vol. 26, pp. 601–609.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Elektrokhimiya, 2017, Vol. 53, No. 4, pp. 407–414.
The article is published in the original.
Rights and permissions
About this article
Cite this article
Chen, Y., Kang, G., Xu, H. et al. PPy doped with different metal sulphate as electrode materials for supercapacitors. Russ J Electrochem 53, 359–365 (2017). https://doi.org/10.1134/S1023193517040036
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193517040036