Abstract
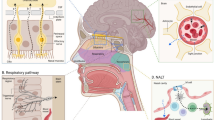
Many nasally applied compounds gain access to the brain and the central nervous system (CNS) with varying degree. Direct nose-to-brain access is believed to be achieved through nervous connections which travel from the CNS across the cribriform plate into the olfactory region of the nasal cavity. However, current delivery strategies are not targeted to preferentially deposit drugs to the olfactory at cribriform. Therefore, we have developed a pressurized olfactory delivery (POD) device which consistently and non-invasively deposited a majority of drug to the olfactory region of the nasal cavity in rats. Using both a hydrophobic drug, mannitol (log P = −3.1), and a hydrophobic drug, nelfinavir (log P = 6.0), and POD device, we compared brain and blood levels after nasal deposition primarily on the olfactory region with POD or nose drops which deposited primarily on the respiratory region in rats. POD administration of mannitol in rats provided a 3.6-fold (p < 0.05) increase in cortex-to-blood ratio, compared to respiratory epithelium deposition with nose drop. Administration of nelfinavir provided a 13.6-fold (p < 0.05) advantage in cortex-to-blood ratio with POD administration, compared to nose drops. These results suggest that increasing the fraction of drug deposited on the olfactory region of the nasal cavity will result in increased direct nose-to-brain transport.








Similar content being viewed by others
References
Newman SP, Pitcairn GR, Dalby RN. Drug delivery to the nasal cavity: in vitro and in vivo assessment. Crit Rev Ther Drug Carrier Syst. 2004;21(1):21–66.
Dhuria SV, Hanson LR, Frey 2nd WH. Intranasal delivery to the central nervous system: mechanisms and experimental considerations. J Pharm Sci. 2010;99(4):1654–73.
Illum L. Is nose-to-brain transport of drugs in man a reality? J Pharm Pharmacol. 2004;56(1):3–17.
Hilger P. Applied anatomy and physiology of the nose. In: Adams GL, LRBaPAH, editors. Boies’s fundamentals of otolaryngology. Philadelphia: W.B. Saunders; 1989. p. 177–95.
Jackson RT, Tigges J, Arnold W. Subarachnoid space of the CNS, nasal mucosa, and lymphatic system. Arch Otolaryngol. 1979;105(4):180–4.
Thorne RG, Frey 2nd WH. Delivery of neurotrophic factors to the central nervous system: pharmacokinetic considerations. Clin Pharmacokinet. 2001;40(12):907–46.
Ross TM, Martinez PM, Renner JC, Thorne RG, Hanson LR, Frey 2nd WH. Intranasal administration of interferon beta bypasses the blood–brain barrier to target the central nervous system and cervical lymph nodes: a non-invasive treatment strategy for multiple sclerosis. J Neuroimmunol. 2004;151(1–2):66–77.
Dahlin M, Jansson B, Bjork E. Levels of dopamine in blood and brain following nasal administration to rats. Eur J Pharm Sci. 2001;14(1):75–80.
Dahlin M, Bergman U, Jansson B, Bjork E, Brittebo E. Transfer of dopamine in the olfactory pathway following nasal administration in mice. Pharm Res. 2000;17(6):737–42.
Jansson B, Bjork E. Visualization of in vivo olfactory uptake and transfer using fluorescein dextran. J Drug Target. 2002;10(5):379–86.
Westin U, Piras E, Jansson B, Bergstrom U, Dahlin M, Brittebo E, et al. Transfer of morphine along the olfactory pathway to the central nervous system after nasal administration to rodents. Eur J Pharm Sci. 2005;24(5):565–73.
Padowski JM, Pollack GM. Examination of the ability of the nasal administration route to confer a brain exposure advantage for three chemical inhibitors of P-glycoprotein. J Pharm Sci. 2010;99(7):3226–33.
Merkus FW, van den Berg MP. Can nasal drug delivery bypass the blood–brain barrier?: questioning the direct transport theory. Drugs R D. 2007;8(3):133–44.
Morrison EE, Costanzo RM. Morphology of the human olfactory epithelium. J Comp Neurol. 1990;297(1):1–13.
Foo MY, Cheng YS, Su WC, Donovan MD. The influence of spray properties on intranasal deposition. J Aerosol Med. 2007;20(4):495–508. Winter.
Illum L. Transport of drugs from the nasal cavity to the central nervous system. Eur J Pharm Sci. 2000;11(1):1–18.
Gross EA, Swenberg JA, Fields S, Popp JA. Comparative morphometry of the nasal cavity in rats and mice. J Anat. 1982;135(Pt 1):83–8.
Thorne RG, Emory CR, Ala TA, Frey 2nd WH. Quantitative analysis of the olfactory pathway for drug delivery to the brain. Brain Res. 1995;692(1–2):278–82.
Liu XF, Fawcett JR, Thorne RG, DeFor TA, Frey 2nd WH. Intranasal administration of insulin-like growth factor-I bypasses the blood–brain barrier and protects against focal cerebral ischemic damage. J Neurol Sci. 2001;187(1–2):91–7.
Hanson LR, Roeytenberg A, Martinez PM, Coppes VG, Sweet DC, Rao RJ, et al. Intranasal deferoxamine provides increased brain exposure and significant protection in rat ischemic stroke. J Pharmacol Exp Ther. 2009;330(3):679–86.
Thorne RG, Pronk GJ, Padmanabhan V, Frey 2nd WH. Delivery of insulin-like growth factor-I to the rat brain and spinal cord along olfactory and trigeminal pathways following intranasal administration. Neuroscience. 2004;127(2):481–96.
Van den Berg MP, Merkus P, Romeijn SG, Verhoef JC, Merkus FW. Hydroxocobalamin uptake into the cerebrospinal fluid after nasal and intravenous delivery in rats and humans. J Drug Target. 2003;11(6):325–31.
Charlton ST, Davis SS, Illum L. Nasal administration of an angiotensin antagonist in the rat model: effect of bioadhesive formulations on the distribution of drugs to the systemic and central nervous systems. Int J Pharm. 2007;338(1–2):94–103.
Young JT. Histopathologic examination of the rat nasal cavity. Fundam Appl Toxicol. 1981;1(4):309–12. Jul–Aug.
Chow HS, Chen Z, Matsuura GT. Direct transport of cocaine from the nasal cavity to the brain following intranasal cocaine administration in rats. J Pharm Sci. 1999;88(8):754–8.
Chow HH, Anavy N, Villalobos A. Direct nose-brain transport of benzoylecgonine following intranasal administration in rats. J Pharm Sci. 2001;90(11):1729–35.
Charlton ST, Whetstone J, Fayinka ST, Read KD, Illum L, Davis SS. Evaluation of direct transport pathways of glycine receptor antagonists and an angiotensin antagonist from the nasal cavity to the central nervous system in the rat model. Pharm Res. 2008;25(7):1531–43.
Cloyd JC, Snyder BD, Cleeremans B, Bundlie SR, Blomquist CH, Lakatua DJ. Mannitol pharmacokinetics and serum osmolality in dogs and humans. J Pharmacol Exp Ther. 1986;236(2):301–6.
Miki K, Butler R, Moore D, Davidson G. Rapid and simultaneous quantification of rhamnose, mannitol, and lactulose in urine by HPLC for estimating intestinal permeability in pediatric practice. Clin Chem. 1996;42(1):71–5.
Williams TF, Hollander Jr W, Strauss MB, Rossmeisl EC, Mc LR. Mechanism of increased renal sodium excretion following mannitol infusion in man. J Clin Invest. 1955;34(4):595–601.
Sisson WB, Oldendorf WH. Brain distribution spaces of mannitol-3H, inulin-14C, and dextran-14C in the rat. Am J Physiol. 1971;221(1):214–7.
Kaddoumi A, Choi SU, Kinman L, Whittington D, Tsai CC, Ho RJ, et al. Inhibition of P-glycoprotein activity at the primate blood–brain barrier increases the distribution of nelfinavir into the brain but not into the cerebrospinal fluid. Drug Metab Dispos. 2007;35(9):1459–62.
Minagar A, Commins D, Alexander JS, Hoque R, Chiappelli F, Singer EJ, et al. NeuroAIDS: characteristics and diagnosis of the neurological complications of AIDS. Mol Diagn Ther. 2008;12(1):25–43.
Hanson LR, Frey 2nd WH. Strategies for intranasal delivery of therapeutics for the prevention and treatment of neuroAIDS. J Neuroimmune Pharmacol. 2007;2(1):81–6.
Jarvis B, Faulds D, Nelfinavir. A review of its therapeutic efficacy in HIV infection. Drugs. 1998;56(1):147–67.
Kandimalla KK, Donovan MD. Localization and differential activity of P-glycoprotein in the bovine olfactory and nasal respiratory mucosae. Pharm Res. 2005;22(7):1121–8.
Graff CL, Pollack GM. P-Glycoprotein attenuates brain uptake of substrates after nasal instillation. Pharm Res. 2003;20(8):1225–30.
Graff CL, Pollack GM. Functional evidence for P-glycoprotein at the nose–brain barrier. Pharm Res. 2005;22(1):86–93.
Acknowledgments
Supported in part by NIH grants AI 077390 and MH086351, and University of Washington Technology Innovation grant TGIF-1001. RJYH is also supported by Milo Gibaldi Endowment.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hoekman, J.D., Ho, R.J.Y. Effects of Localized Hydrophilic Mannitol and Hydrophobic Nelfinavir Administration Targeted to Olfactory Epithelium on Brain Distribution. AAPS PharmSciTech 12, 534–543 (2011). https://doi.org/10.1208/s12249-011-9614-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9614-1