Abstract
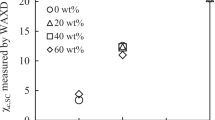
High-molecular-weight poly(L-lactic acid-co-glycolic acid) (PLGA) was prepared through melt polymerization from glycolic acid (GA) and L-lactic acid (L-LA) and then characterized. High resolution 1H and 13C NMR were performed for microstructure analysis of polymer. The solubility in chloroform, compositions, and sequence lengths of PLGA suggest higher reactivity of GA compared with L-LA and shortening GA blocks with reaction time. The racemization of L-LA blocks and transesterification in PLGA are enhanced by increase of GA fractions. PLGA (90/10) samples show crystallization of relatively long L-LA sequences in copolymer chains and the absence of the crystallization of GA blocks. All these results demonstrate the influence of racemization and transesterification on the microstructure of PLGA.
Similar content being viewed by others
Article PDF
References
D. W. Grijpma, G. J. Zondervan, and A. J. Pennings, Polym. Bull., 25, 327 (1991).
D. W. Grijpma and A. J. Pennings, Polym. Bull., 25, 335 (1991).
G. Chen, T. Ushida, and T. Tateishi, Biomaterials, 22, 2563 (2001).
M. Dunne, O. I. Corrigan, and Z. Ramtoola, Biomaterials, 21, 1659 (2000).
W. H. Carothers, G. L. Dorough, and F. J. Van Na, J. Am. Chem. Soc., 54, 761 (1932).
H. R. Kricheldorf and I. Kreiser, Makromol. Chem., 188, 1861 (1987).
J. Kasperczyk, Polymer, 37, 201 (1996).
D. W. Grijpma, A. J. Nijenhuis, and A. J. Pennings, Polymer, 31, 2201 (1990).
S. M. F. Farnia, J. Mohammadi-Rovshandeh, and M. N. Sarabolouki, J. Appl. Polym. Sci., 73, 633 (1999).
D. D. Hile and M. V. Pishko, Makromol. Rapid Commun., 20, 511 (1999).
D. D. Hile and M. V. Pishko, J. Polym. Sci., Part A: Polym. Chem., 39, 562 (2001).
D. Cohn, H. Younes, and G. Marom, Polymer, 28, 2018 (1987).
D. K. Gilding and A. M. Reed, Polymer, 20, 1459 (1979).
D. K. Gilding and A. M. Reed, Polymer, 22, 494 (1981).
H. Fukuzaki, M. Yoshida, M. Asano, Y. Aiba, and M. Kumakura, Eur. Polym. J., 26, 457 (1990).
N. Wang, X. S. Wu, H. Lujian-Upton, E. Donahue, and A. Siddiqui, Polym. Mater. Sci. Eng., 76, 373 (1997).
J. L. Espartero, I. Rashkov, S. M. Li, N. Manolova, and M. Vert, Macromolecules, 29, 3535 (1996).
H. Fukuzaki, M. Yoshida, M. Asano, and Y. Aiba, Eur. Polym. J., 24, 1029 (1988).
K. Imasaka, T. Nagai, M. Yoshida, H. Fukuzaki, M. Asano, and M. Kumakura, Eur. Polym. J., 26, 831 (1990).
K. Imasaka and T. Nagai, Makromol. Chem., 191, 2077 (1990).
H. Fukuzaki, M. Yoshida, M. Asano, and M. Kumakura, Eur. Polym. J., 25, 1019 (1989).
M. Ajioka, K. Enomoto, K. Suzuki, and A. Yamaguchi, Bull. Chem. Soc. Jpn., 68, 2125 (1995).
M. Ajioka, K. Suizu, C. Higuchi, and T. Kashima, Polym. Degrad. Stab., 59, 137 (1998).
S. I. Moon, C. W. Lee, M. Miyamoto, and Y. Kimura, J. Polym. Sci., Part A: Polym. Chem., 38, 1673 (2000).
S. I. Moon, C. W. Lee, I. Taniguchi, M. Miyamoto, and Y. Kimura, Polymer, 42, 5059 (2001).
K. Takahashi, I. Taniguchi, M. Miyamoto, and Y. Kimura, Polymer, 41, 8725 (2000).
Von R. C. Schukz and J. Schaab, Makromol. Chem., 87, 90 (1965).
T. Miyata and T. Masuko, Polymer, 39, 5515 (1998).
E. J. Choi, J. K. Park, and H. N. Chang, J. Polym. Sci., Part B: Polym. Phys., 32, 2481 (1994).
A. Schindler and D. Harper, J. Polym. Sci., Polym. Lett. Ed., 14, 72 (1976).
J. Kasperczyk and M. Bero, Makromol. Chem., 194, 913 (1993).
M. Bero, J. Kasperczyk, and G. Adamus, Makromol. Chem., 194, 907 (1993).
J. Kasperczyk and M. Bero, Makromol. Chem., 192, 1777 (1991).
M. Bero, J. Kasperczyk, and Z. J. Jedlinski, Makromol. Chem., 191, 2287 (1990).
P. Vanhoorne, Ph. Dubois, R. Jerome, and P. Teyssie, Macromolecules, 25, 37 (1992).
E. Lillie and R. C. Schulf, Makromol. Chem., 176, 1901 (1975).
Z. Jedlinski and W. Walach, Makromol. Chem., 192, 2051 (1991).
N. Spassky, M. Wisniewski, C. Pluta, and A. L. Borgne, Macromol. Chem. Phys., 197, 2627 (1996).
J. Coudane, C. U. Peyret, G. Schwach, and M. Vert, J. Polym. Sci., Part A: Polym. Chem., 35, 1651 (1997).
F. Chabot, M. Vert, S. Chapelle, and P. Granger, Polymer, 24, 53 (1983).
K. A. M. Thakur, P. T. Kean, E. S. Hall, J. J. Kolstad, T. A. Lindgren, M. A. Doscotch, J. I. Siepmann, and E. J. Munson, Macromolecules, 30, 2422 (1997).
E. W. Fischer, H. J. Sterzel, and G. Wegner, Kolloid-Z. u. Z. Polymere, 251, 980 (1973).
H. R. Kricheldorf, C. Boettcher, and K. U. Tonnes, Polymer, 33, 2817 (1992).
D. Brizzolara and H.-J. Cantow, Macromolecules, 29, 191 (1996).
W. Hoogsteen, A. R. Postema, A. J. Pennings, G. Ten Brinke, and P. Zugenmaier, Macromolecules, 23, 634 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gao, Q., Lan, P., Shao, H. et al. Direct Synthesis with Melt Polycondensation and Microstructure Analysis of Poly(L-lactic acid-co-glycolic acid). Polym J 34, 786–793 (2002). https://doi.org/10.1295/polymj.34.786
Issue Date:
DOI: https://doi.org/10.1295/polymj.34.786
Keywords
This article is cited by
-
Characterization of commercial PLGAs by NMR spectroscopy
Drug Delivery and Translational Research (2022)
-
Well-defined high molecular weight polyglycolide-b-poly(L-)lactide-b-polyglycolide triblock copolymers: synthesis, characterization and microstructural analysis
Journal of Polymer Research (2020)
-
Synthesis of Bioresorbable Poly(Lactic-co-Glycolic Acid)s Through Direct Polycondensation: An Economical Substitute for the Synthesis of Polyglactin via ROP of Lactide and Glycolide
Fibers and Polymers (2019)
-
Synthesis and Characterization of Poly(ester amide)s Consisting of Poly(L-lactic acid) and Poly(butylene succinate) Segments with 2,2′-Bis(2-oxazoline) Chain Extending
Macromolecular Research (2018)
-
Synthesis of PLGA using a C 3-symmetric Zr (IV) amine tris(phenolate) alkoxide initiator and the effects of gamma radiation on its properties
Polymer Bulletin (2017)