Abstract
A correlation was made of crystallochemical data from 15 smectites with the half time of dissolution (t05) in 6 N hydrochloric acid at 96°C. From this the relation
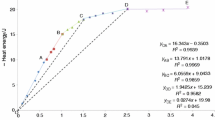
ln t05 = 3.95 - 1.96 FeVI - 2.30 MgVI
was developed. It shows a remarkable dependence of the apparent dissolution rate of the octahedral layer of the smectites on the substitution of Fe3+ and Mg2+ for Al+3 in the octahedral position.
Резюме
Было проведено изучение корреляционной зависимости между кристалло-химическими данными 15 образцов смектита и половиной времени их растворения (t05) в 6N соляной кислоте при 9б°С. Отсюда было получено следующее соотношение: ln t05=3.95-1.96FeVI-2.30 MgVI. Оно показывает примечательную зависимость удельной скорости растворения октаэдрического слоя смектитов от замещения Al+3 катионами Fe3+ и Mg2+ в октаэдрической позиции.
Kurzreferat
Eine Korrelation kristallchemischer Daten von 15 Smektiten mit den Halbwertszeiten der Auflösung (t05) in 6N HCl bei 96°C wurde aufge-stellt. Daraus wurde die Gleichung: ln t05=3,95-1,96 FeVI-2,30 MgVI abgeleitet. Es zeigte sich eine bemerkenswerte Abhängigkeit der scheinbaren Auflösungsrate der oktahedrischen Schicht der Smektiten von der Substitution von Fe(III) und Mg(II) für Al(III) in der oktahedrischen Position.
Résumé
Une corrélation a été établie entre les données cristallochimiques de quinze smectites ayant un demi-temps de dissolution(t05) dans l’acide hydrochlorique 6N à 96°C.La relation suivante a été développée: ln t05=3.95-1.96 FeVI-2.30 MgVI
Elle montre une dépendance remarquable de la vitesse apparente de dissolution du feuillet octaèdral des smectites de la substitution de Fe3+ et Mg2+ à Al+3 dans la position octaèdrale.
Similar content being viewed by others
References
Brindley, G. W. and Youell, F. (1951) A chemical determination of the tetrahedral and octahedral aluminium ions in a silicate: Acta Crystallogr. 4, 495–497.
Číčel, B. and Novák, I. (1976) Dissolution of smectites in HCl: I. Half-time of dissolution as a measure of reaction rate: Proc. 7th Conf. Clay Mineral. Petrol, (in print), Charles Univ., Prague.
Číčel, B., Novák, I. and Pivovarníček, F. (1965) Dissolution of mont-morillonite in HCl and its possible application in the study of their activation: Silikáty 9, 130–140 (Slovak).
Donnay, G., Donnay, J. D. H. and Takeda, H. (1964) Trioctahedral one-layer micas. II. Prediction of the structure from composition and cell dimensions: Acta Crystallogr. 17, 1374–81.
Ezekiel, M. and Fox, K. A. (1959) Methods of Correlation and Regression Analysis. Wiley Inc., New York.
Fahn, R. (1963) Innerkristalline Quellung und Farbstoffadsorption säurebehandelter Montmorillonite: Kolloid Z. 187, 120–127.
Granquist, W. T. and Gardner-Sumner, G. (1959) Acid dissolution of the Texas bentonite: Clays & Clay Minerals 6, 292–308.
Gregor, M., Číčel, B. and Krempaský, V. (1966) Kinetische Studie des Zerfalles von Montmorillonit in Salzsàure: Zb. Pr. Chemickotechnol. Fak. SVST 91–96.
Karšulin, M. and Stubičan, V. (1954) Über die Struktur und die Eigenschaften synthetischer Montmorillonite: Monatsh. Chem. 85, 343–358.
Osthaus, B. B. (1954) Chemical determination of terahedral ions in nontronite and montmorillonite: Clay & Clay Minerals 2, 404–417.
Osthaus, B. B. (1956) Kinetic studies on montmorillonites and nontronite by acid dissolution technique: Clays & Clay Minerals 4, 301–321.
Packter, A. (1955) Catalysis by complexing anions. The kinetics of the activation of montmorillonite clays: Chem. Ind. 1384.
Turner, R. (1964) Kinetic studies of acid dissolution of montmorillonite and kaolinite: Ph.D. Thesis Univ. Calif.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Novák, I., Číčel, B. Dissolution of Smectites in Hydrochloric Acid: II. Dissolution Rate as a Function of Crystallochemical Composition. Clays Clay Miner. 26, 341–344 (1978). https://doi.org/10.1346/CCMN.1978.0260504
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1978.0260504