Abstract
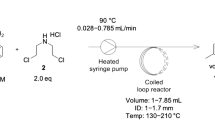
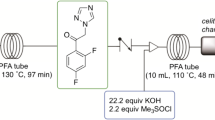
The use of continuous-flow chemistry has shown to be an important tool in improving API manufacture. In the present paper, we report the use of continuous-flow reactors in the synthesis of the bicyclic side chain of antiretroviral Darunavir.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Microreactors; Ehrfeld, W.; Hessel, V.; Löwe, H., Eds.; Wiley-VCH: Weinheim, 2000
Chemical MicroProcess Engineering; Hessel, V.; Hardt, S.; Löwe, H., Eds.; Wiley-VCH: Weinheim, 2004
Microreactors in Organic Synthesis; Wirth, T., Ed., Wiley-VCH: Weinheim, 2008
Flash Chemistry: Fast Organic Synthesis in Microsystems, Yoshida, J., Ed.; Wiley-Blackwell: Oxford, 2008
MicroProcess Engineering, Hessel, V.; Renken, A.; Schouten, J. C.; Yoshida, J., Eds.; Wiley-Blackwell: Oxford, 2009.
For selected recent reviews on continuous-flow/microreactor chemistry, see: (a) Wiles, C. Watts, P. Green Chem. 2012, 14, 38–54
Noël, T.; Buchwald, S. L. Chem. Soc. Rev. 2011, 40, 5010–5029
Baumann, M.; Baxendale, I. R.; Ley, S. V. Mol. Diversity 2011, 15, 613–630
Hartman, R. L.; McMullen, J. P.; Jensen, K. F. Angew. Chem., Int. Ed. 2011, 50, 7502–7519
Wiles, C.; Watts, P. Chem. Commun. 2011, 47, 6512–6535.
Wegner, J.; Ceylan, S.; Kirschning, A. Chem. Commun. 2011, 47, 4583–4592.
Yoshida, J. I.; Kim, H.; Nagaki, A. ChemSusChem 2011, 4, 331–340.
Wegner, J.; Ceylan, S.; Kirschning, A. Adv. Synth. Catal. 2012, 354, 17–57
Jimenez-Gonzalez, C.; Poechlauer, P.; Broxterman, Q. B.; Yang, B. S.; am Ende, D.; Baird, J.; Bertsch, C.; Hannah, R. E.; Dell’Orco, P.; Noorman, H.; Yee, S.; Reintjens, R.; Wells, A.; Massonneau, V.; Manley, J. Org. Process Res. Dev. 2011, 15, 900–911.
For selected examples of API synthesis in flow: (a) Hopkin, M. D.; Baxendale, I. R.; Ley, S. V. Chem. Commun. 2010, 46, 2450–2452
Grongsaard, P.; Bulger, P. G.; Wallace, D. J.; Tan, L.; Chen, Q.; Dolman, S. J.; Nyrop, J.; Hoerrner, R. S.; Weisel, M.; Arredondo, J.; Itoh, T.; Xie, C.; Wen, X.; Zhao, D.; Muzzio, D. J.; Bassan, E. M.; Shultz, C. S Org. Process Res. Dev. 2012, 16, 1069–1081
Gustafsson, T.; Sörensen, H.; Ponten, F. Org. Process Res. Dev. 2012, 16, 925–929
Johnson, M. D.; May, S. A.; Calvin, J. R.; Remacle, J.; Stout, J. R.; Diseroad, W. D.; Zaborenko, N.; Haeberle, B. D.; Sun, W. M., Miller, M. T.; Brennan, J. Org. Process Res. Dev. 2012, 16, 1017–1038
Hopkin, M. D.; Baxendale, I. R.; Ley, S. V. Org. Biomol. Chem. 2013, 11, 1822–1839
Gutmann, B.; Cantillo, D.; Kappe, C. O. Angew. Chem. Int. Ed. 2015, 54, 6688.
Della-Vechia, L.; Reichard, B.; Glasnov, T.; Miranda, L. S. M.; Kappe, C. O.; Souza, R. O. M. A. Org. Biomol. Chem. 2013, 11, 6806–6813
Pinho, V. D.; Gutmann, B.; Miranda, L. S. M.; Souza, R. O. M. A.; Kappe, C. O. J. Org. Chem. 2014, 79, 1555–1562.
Ebhralm, O.; Mazanderano, A. H. Infect. Dis. Rep. 2013, 5, Suppl 1, e2
Eholië, S. P.; Aossi, F. E.; Ouattara, I. S.; Bissagnënë, E.; Anglaret, X. J. Int. AIDS Soc. 2012, 15, 17334–17340.
Ghosh, A. K.; Dawsona, Z. L.; Mitsuya, H. Bioorg. Med. Chem. 2007, 15, 7576–7580.
Yu, R. H.; Polniaszek, R. P.; Becker, M. W.; Cook, C. M.; Yu, L. H. L. Org. Process. Res. Dev., 2007, 11, 972–980
Black, D. M.; Davis, R.; Doan, B. D.; Lovelace, T. C.; Millar, A.; Toczko, J. F.; Xie, S. Tetrahedron: Asymmetry, 2008, 19, 2015–2019
Ghosh, A. K.; Kincaid, J. F.; Walters, D. E. J. Med. Chem. 1996, 39, 3278–3290
Ghosh, A. K.; Li, J.; Perali, R. S. Synthesis 2006, 3015–3018
Ghosh, A. K.; Leshchenko, S.; Noetzel, M. J. Org. Chem. 2004, 69, 7822–7829
Ghosh, A. K.; Chen, Y. Tetrahedron Lett. 1995, 36, 505–508.
Quaedflieg, P. J. L. M.; Kesteleyn, B. R. R.; Wigerinck, P. B. T. P.; Goyvaerts, N. M. F.; Vijn, R. J.; Liebregts, C. S. M.; Kooistra, J. H. M. H.; Cusan, C. Org. Lett. 2005, 7, 5917–5920
Canoy, W. L.; Cooley, B. E.; Corona, J. A.; Lovelace, T. C.; Millar, A.; Weber, A. M.; Xie, S.; Zhang, Y. Org. Lett. 2008, 10, 1103–1106.
de Miranda, A. S.; Miranda, L. S. M.; Souza, R. O. M. A. Org. Biomol. Chem., 2013, 11, 3332–3336
de Miranda, A. S.; Miranda, L. S. M.; Souza, R. O. M. A. RSC Adv., 2014, 4, 13620–13625
de Miranda, A. S.; Gomes, J. C.; Rodrigues, M. T; Costa, I. C. R.; Almeida, W. P.; Lopes, R. O.; Miranda, L. S. M.; Coelho, F.; Souza, R. O. M. A. J. Mol. Cat. B: Enzymatic 2013, 91, 77–80.
Khmelnitsky, Y. L.; Michels, P. C.; Cotterill, I. C.; Eissenstat, M.; Sunku, V.; Veeramaneni, V. R.; Cittineni, H.; Kotha, G. R.; Talasani, S. R.; Ramanathan, K. K.; Chitineni, V. K.; Venepalli, B. R. Org. Process. Res. Dev. 2011, 15, 279–283.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Leão, R.A.C., de Lopes, R.O., Bezerra, M.A.d. et al. Studies on the Continuous-Flow Synthesis of Nonpeptidal bis-Tetrahydrofuran Moiety of Darunavir. J Flow Chem 5, 216–219 (2015). https://doi.org/10.1556/1846.2015.00031
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/1846.2015.00031