Abstract
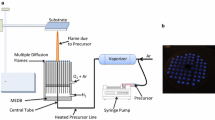
A counterflow diffusion flame burner was used to produce nanophase vanadium-phosphorus oxide powders in a hydrogen-oxygen flame. Liquid precursors, i.e., VOCl3 and PCl3, were used as source materials in a 1:1 ratio. In situ formation processes were investigated at two temperatures by laser light scattering, by emission and absorption spectroscopy, and by collecting particles directly onto carbon-coated TEM grids. At the higher temperature, the collected powders are spherical particles about 30 to 50 nm in diameter. At the lower temperature, the powders collected are chain-like structures composed of particles 5 to 10 nm in diameter. Particles formed in the burner were collected also from the burner’s flanges and from two auxiliary strips. Their crystalline phases and surface area were determined by x-ray diffractometry, FT-IR spectroscopy, and BET analysis by nitrogen desorption. These results indicate a strong influence of temperature on the crystalline phases of the powders. At the higher temperature, the powder collected is a mixture of VOPO4 · 2H2O and δ-VOPO4. This mixture forms Λ-VOPO4 upon subsequent reheating at 750 °C. At the lower temperature, the powders collected are a VOHxPO4 · yH2O phase and VO(H2PO4)2, and form β-VOPO4 and V(PO3)3, respectively, upon subsequent reheating at 750 °C.
Similar content being viewed by others
References
M. Ai, P. Boutry, and R. Montarnal, Bull. Soc. Chim. Fr. 8, 2775 (1970).
M. Ai, P. Boutry, R. Montarnal, and G. Thomas, Bull. Soc. Chim. Fr. 8, 2783 (1970).
E. Bordes, Catal. Today 16, 27 (1993).
E. Bordes, Catal. Today 1, 499 (1987).
B.K. Hodnett, Catal. Rev.-Sci. Eng. 27, 373 (1985).
G. Busca, F. Cavani, G. Centi, and F. Trifirò, J. Catal. 99, 400 (1986).
E. Bordes and P. Courtine, J. Chem. Soc, Chem. Commun. 294 (1985).
E. Bordes and P. Courtine, J. Catal. 57, 236 (1979).
N. Harrouch Batis, H. Batis, A. Ghorbel, J. C. Vedrine, and J. C. Volta, J. Catal. 128, 248 (1991).
P. Courtine, Solid State Chemistry in Catalysis, A.C.S. Symp. Series 279, 37 (1985).
J.C. Volta and J.L. Portefaix, Appl. Catal. 18, 1 (1985).
E. Bordes, P. Courtine, and G. Pannetier, Ann. Chim. 8, 105 (1973).
B. K. Hodnett and B. Delmon, Appl. Catal. 9, 203 (1984).
G. Centi, I. Maneti, A. Riva, and F. Trifirò, Appl. Catal. 9, 177 (1984).
G. Poli, I. Resta, O. Ruggeri, and F. Trifirò, Appl. Catal. 1, 395 (1981).
F. Cavani, G. Centi, and F. Trifirò, Appl. Catal. 9, 191 (1984).
J. W. Johnson, D. C. Johnston, and A. J. Jacobson, in Preparation of Catalysts IV (Elsevier Science Publishing, Amsterdam, 1987).
E. Bordes, J. W. Johnson, A. Raminosona, and P. Courtine, Mater. Sci. Monograf. 28B, 887 (1985).
J.W. Johnson, D.J. Johnston, A.J. Jacobson, and J.F. Brody, J. Am. Chem. Soc. 106, 8123 (1984).
D. D. Beck and R. W. Siegel, J. Mater. Res. 7, 2840 (1992).
C-H. Hung and J.L. Katz, J. Mater. Res. 7, 1861 (1992).
C-H. Hung, P. F. Miquel, and J. L. Katz, J. Mater. Res. 7, 1870 (1992).
P.F. Miquel, C-H. Hung, and J.L. Katz, J. Mater. Res. 8, 2404 (1993).
J. L. Katz and C-H. Hung, Combust. Sci. Technol. 82, 169 (1992).
H.J. Kostkowski and H.P. Broida, J. Opt. Soc. Am. 46, 246 (1956).
G. H. Dieke and H. M. Crosswhite, J. Quant. Spectrosc. Radiat. Transfer 2, 97 (1962).
S.L. Chung, Ph.D. Thesis, The Johns Hopkins University, Baltimore, MD (1985).
A. G. Gaydon, The Spectroscopy of Flame (Chapman and Hall, London, 1974).
R. M. Dagnall, K. C. Thompson, and T. S. West, Analyst 93, 72 (1968).
C-H. Hung, Ph.D. Thesis, The Johns Hopkins University, Baltimore, MD (1991).
R. A. Dobbins and CM.. Megaridis, Langmuir 3, 254 (1987).
G. V. Samsonov, The Oxide Handbook (Plenum Press, New York, 1973).
The chain-like and the spherical particles collected on TEM grids in Flame 1 have structures and morphologies similar to those collected in Flame 2 (shown in Fig. 4), although their sizes are different, as stated in the text.
G. Ladwig, Z. Anorg. Allg. Chem. 338, 266 (1965).
A. Schneider, Thesis, Université de Bordeaux, France (1987).
R.N. Bhargava and R.A. Condrate, Appl. Spectrosc. 3, 230 (1977).
P. Amorós, R. Ibáñez, E. Martinez-Tamayo, A. Beltrán-Porter, and D. Beltrân-Porter, Mat. Res. Bull. XXIV, 1347 (1989).
G. Ladwig, Z. Chem. 8, 307 (1968).
Note that x-ray and FT-IR analyses of the powders collected in both Flames show the presence of hydrated phases. Because of the high temperatures, it is likely that the powders which actually formed in the flame are not hydrated, and that these highly hygroscopic powders hydrated on the collection strips or in later handling.
A. V. Lavrov, L. S. Guzeeva, and P. M. Fedorov, Izv. Akad. Nauk SSSR, Neorg. Mater. 10, 1280 (1974).
B.C. Tofield, G.R. Crane, G.A. Pasteur, and R. C. Sherwood, J. Chem. Soc. Dalton Trans, 1806 (1975).
Author information
Authors and Affiliations
Additional information
Address all correspondence to this author.
Rights and permissions
About this article
Cite this article
Miquel, P.F., Katz, J.L. Formation and characterization of nanostructured V—P—O particles in flames: A new route for the formation of catalysts. Journal of Materials Research 9, 746–754 (1994). https://doi.org/10.1557/JMR.1994.0746
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1994.0746