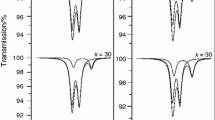
Abstract
The conductivity of the Li2O:P2O5:MO3 (M = Cr2, Mo, W) glasses increases as P2O5 is progressively substituted by MO3 and as the Li2O content increases. Amongst the glass compositions studied, the 0.50Li20:0.20P2O5:0.30WO3 glass has the highest conductivity at 25°C of 2.1×10−6 Ω−1cm−1. The glass transition temperature of the glasses increases initially with network former substitution, reaches a maximum at around MO3/P2O5 = 1, and decreases with further substitution. X-ray photoelectron spectroscopy reveals the presence of M ions in more than one oxidation state and oxygen species such as P=O, P-O-P, P-O, M-O-M, M-O and P-O-M. Raman spectroscopy shows that the Li2O:P2O5:MoO3 and Li2O:P2O5:WO3 glasses consist of PO4, MoO4 (WO4) and MoO6 (WO6) polyhedra while the Li2O:P2O5:Cr2O3 glasses consist of the PO4 and CrO6 polyhedra only. The phosphate groups are preferentially modified by Li2O in comparison with the tungstate, molybdate and chromate groups. The increasing number of non-bridging oxygen atoms per phosphate group may be related to the increasing conductivity with the progressive substitution of MO3 for P2O5.
Similar content being viewed by others
References
B.V.R. Chowdari, K.L. Tan, W.T. Chia and R. Gopalakrishnan, J. Non-Cryst. Solids 128, 18 (1991).
B.V.R. Chowdari, K.L. Tan, W.T. Chia and R. Gopalakrishnan, in Recent Advances in Fast Ion Conducting Materials and Devices, edited by B.V.R. Chowdari, Q.G. Liu and L.Q. Chen (World Scientific, Singapore, 1990), p. 513.
B.V.R. Chowdari, K.L. Tan and W.T. Chia, Solid State Ionics 53–56, 1172 (1992).
W.T. Chia, B.V.R. Chowdari and K.L. Tan, to be published in J. Mater. Sci..
A. Eisenberg, H. Farb and L.G. Cool, J. Polymer Sci. A-2 4, 855 (1966).
S.W. Martin and C.A. Angell, J. Non-Cryst. Solids 83, 185 (1986).
S.W. Martin and C.A. Angell, J. Phys. Chem. 90, 6736 (1986).
K. Nassau, A.M. Glass, M. Grasso and D.H. Olson, J. Electrochem. Soc. 127, 2743 (1980).
B.V.R. Chowdari, K.L. Tan, W.T. Chia and R. Gopalakrishnan, J. Non-Cryst. Solids 119, 95 (1990).
M. Tatsumisago, Y. Kowada and T. Minami, Phys. Chem. Glasses 29, 63 (1988).
I.R. Beattie and T.R. Gilson, J. Chem. Soc. A 1969, 2322.
C.G. Hill, Jr. and J.H. Wilson III, J. Molecular Catalysis 63, 65 (1990).
S.H. Morgan and R.H. Magruder III, J. Am. Ceram. Soc. 73, 753 (1990).
R.H. Busey and O.L. Keller, Jr., J. Chem. Phys. 41, 215 (1964).
F.L. Galeener and J.C. Mikkelsen, Jr., Solid St. Commun. 30, 505 (1979).
P. Kierkegaard, K. Eistrat and A.R. Rosenhall, Acta. Chem. Scand. 18, 2237 (1964).
A.F. Wells, Structural Inorganic Chemistry (Oxford, Oxford, 1984), p. 595.
T. Yokokawa and S. Kohsaka, J. Chem. Eng. Data 24, 167 (1979).
Y. Kowada, M. Tatsumisago and T. Minami, J. Phys. Chem. 93, 2147 (1989).
A. Pradel, T. Pagnier and M. Ribes, Solid State Ionics 17, 147 (1985).
M. Scagliotti, M. Villa and G. Chiodelli, J. Non-Cryst. Solids 93, 350 (1987).
M. Villa, M. Scagliotti and G. Chiodelli, J. Non-Cryst. Solids 94, 101 (1987).
T. Furukawa and W.B. White, J. Mater. Sci. 16, 2689 (1981).
C. Nelson and D.R. Tallant, Phys. Chem. Glasses 25, 31 (1984).
I.N. Chakraborty and R.A. Condrate, Snr., Phys. Chem. Glasses 26, 68 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chowdari, B., Tan, K. & Chia, W. Structural and Physical Characterization of Li2O:P2O5:MO3 (M = Cr2, Mo, W) Ion Conducting Glasses. MRS Online Proceedings Library 293, 325–336 (1992). https://doi.org/10.1557/PROC-293-325
Published:
Issue Date:
DOI: https://doi.org/10.1557/PROC-293-325