Abstract
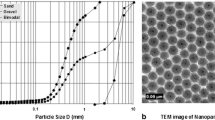
In this study, the transport of water-stable “nano-C60 particles” (a term used to refer to underivatized C60 crystalline nanoparticles, stable in water for months) through a soil column (packed with Lula soil, 0.27% organic carbon) was investigated for the first time. Nano-C60 particle breakthrough experiments were conducted at different flow rates, while other column operating parameters remained fixed through all the experiments. Nano-C60 particles were observed to be more mobile at higher flow velocity: at the flow velocity of 0.38 m/d, the maximum percent of nano-C60 breakthrough (C/C0) was 47%; at the flow velocity of 3.8 m/d, the plateau value of nano-C60 breakthrough was 60%; and at the flow velocity of 11.4 m/d, the plateau value of nano-C60 breakthrough was almost 80%. At the low flow velocity (0.38 m/d), which is typical of groundwater flow, nano-C60 particles showed very limited mobility: after about 57 pore volumes, they deposited to the soil column so rapidly that virtually no nano-C60 was detected in the effluent. This observed “favorable deposition” (attachment efficiency α = 1) was probably due to “filter ripening.” Also the release of nano-C60 particles after flow interruption was observed. The transport of naphthalene through the same soil column containing 0.18% nano-C60 particles deposited was measured. A retardation factor of about 13 was observed, possibly suggesting that sorbed nano-C60 particles in the soil column sorbed naphthalene similar to soil organic carbon. An asymmetric naphthalene breakthrough curve was observed, which is possibly due to “sorption nonequilibrium.”
Similar content being viewed by others
References
V.L. Colvin: The potential environmental impact of engineered nanomaterials. Nat. Biotechnol. 21 1166 (2003).
R.S. Ruoff D.S. Tse R. Malhotra and D.C. Lorents: Solubility of fullerene (C60) in a variety of solvents. J. Phys. Chem. 97 3379 (1993).
W.A. Scrivens J.M. Tour K.E. Creek and L. Pirisi: Synthesis of 14C-labeled C60 its suspension in water and its uptake by human keratinocytes. J. Am. Chem. Soc. 116 4517 (1994).
G.V. Andrievsky M.V. Kosevich O.M. Vovk V.S. Shelkovsky and L.A. Vashchenko: On the production of an aqueous colloidal solution of fullerenes. J. Chem. Soc. Chem. Commun. 12 1281 (1995).
S. Deguchi R.G. Alargova and K. Tsujii: Stable dispersions of fullerenes C60 and C70 in water. Preparation and characterization. Langmuir 17 6013 (2001).
E. Oberdorster: Manufactured nanomaterials (fullerenes C60) induce oxidative stress in the brain of juvenile largemouth bass. Environ. Health Perspect. 112 1058 (2004).
C.M. Sayes J.D. Fortner W. Guo D. Lyon A.M. Boyd K.D. Ausman Y.J. Tao B. Sitharaman L.J. Wilson J.B. Hughes J.L. West and V.L. Colvin: The differential cytotoxicity of water-soluble fullerenes. Nano Lett. 4 1881 (2004).
A.T. Kan and M.B. Tomson: Ground water transport of hydrophobic organic compounds in the presence of dissolved organic matter. Environ. Toxicol. Chem. 9 253 (1990).
H.F. Lecoanet J. Bottero and M.R. Wiesner: Laboratory assessment of the mobility of nanomaterials in porous media. Environ. Sci. Technol. 38 5164 (2004).
H.F. Lecoanet and M.R. Wiesner: Velocity effects on fullerene and oxide nanoparticle deposition in porous media. Environ. Sci. Technol. 38 4377 (2004).
J.T. Wilson C.G. Enfield W.J. Dunlap R.L. Cosby D.A. Foster and L.B. Baskin: Transport and fate of selected organic pollutants in a sandy soil. J. Environ. Qual. 10 501 (1981).
G.L. Clark: Flow rate effects on the sorption of methylated benzenes in saturated aquifer materials. Ph.D. Dissertation Rice University Houston TX (1990).
X. Cheng A.T. Kan and M.B. Tomson: Uptake and sequestration of naphthalene and 1,2-dichlorobenzene by C60. J. Nanoparticle Res. 7 555 (2005).
K.M. Yao M.T. Habibian and O’C.R. Melia: Water and wastewater filtration: Concepts and applications. Environ. Sci. Technol. 5 1105 (1971).
O’C.R. Melia: Kinetics of colloid chemical processes in aquatic systems in Aquatic Chemical Kinetics–Reaction Rates of Processes in Natural Waters edited by W. Stumm (John Wiley & Sons New York 1990) pp. 447–474.
R. Rajagopalan and C. Tien: Trajectory analysis of deep-bed filtration with the sphere-in-cell porous media model. Am. Inst. Chem. Eng. J. 22 523 (1976).
R.W. Harvey L.H. George R.L. Smith and D.R. Leblanc: Transport of microspheres and indigenous bacteria through a sandy aquifer: Results of natural and forced-gradient tracer experiments. Environ. Sci. Technol. 23 51 (1989).
J.E. Tobiason and C.R. O’Melia: Physicochemical aspects of particle removal in depth filtration. J. Am. Water Works Assoc. 80 54 (1988).
R. Kretzschmar and H. Sticher: Transport of humic-coated iron oxide colloids in a sandy soil: Influence of Ca2+ and trace metals. Environ. Sci. Technol. 31 3497 (1997).
M. Elimelech J. Gregory X. Jia and R.A. Williams: Particle Deposition and Aggregation. Measurement Modeling and Simulation (Butterworth-Heinemann Woburn MA 1995).
M. Elimelech and O’C.R. Melia: Kinetics of deposition of colloidal particles in porous media. Environ. Sci. Technol. 24 1528 (1990).
D. Grolimund M. Elimelech M. Borkovec K. Barmettler R. Kretzschmar and H. Sticher: Transport of in situ mobilized colloidal particles in packed soil columns. Environ. Sci. Technol. 32 3562 (1998).
J. Zhuang Y. Jin and M. Flury: Comparison of Hanford colloids and kaolinite transport in porous media. Vadose Zone J. 3 395 (2004).
J. Bear: Hydraulics of Groundwater (McGraw-Hill New York 1979).
A.T. Kan and M.B. Tomson: Effect of pH concentration on the transport of naphthalene in saturated aquifer media. J. Contam. Hydrol. 5 235 (1990).
J.N. Wilson: A theory of chromatography. J. Am. Chem. Soc. 62 1583 (1940).
M.L. Brusseau and P.S.C. Rao: Sorption nonideality during organic contaminant transport in porous media. Crit. Rev. Environ. Control. 19 33 (1989).
J.C. Giddings and H. Eyring: A molecular dynamic theory of chromatography. J. Phys. Chem. 59 416 (1955).
P.S.C. Rao R.E. Green V. Balasubramanian and Y. Kanehiro: Fielf study of solute movement in a highly aggregated oxisol with intermittent flooding. II. Picloram. J. Environ. Qual. 3 197 (1974).
X. Cheng A.T. Kan and M.B. Tomson: Naphthalene adsorption and desorption from aqueous C60 fullerene. J. Chem. Eng. Data 49 675 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cheng, X., Kan, A.T. & Tomson, M.B. Study of C60 transport in porous media and the effect of sorbed C60 on naphthalene transport. Journal of Materials Research 20, 3244–3254 (2005). https://doi.org/10.1557/jmr.2005.0402
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2005.0402