Abstract
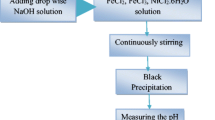
In this research work, we prepared γ-Fe2O3 nanoparticles by thermal-decomposition of Fe3O4. The Fe3O4 nanoparticles were synthesized via co-precipitation method at room temperature. This simple, soft and cheap method is suitable for preparation of iron oxide nanoparticles (γ-Fe2O3; Fe3O4). The samples were characterized by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), transmission electron microscopy (TEM), vibrating sample magnetometer and differential scanning calorimeter (DSC). The XRD and FT-IR results indicated the formation of γ-Fe2O3 and Fe3O4 nanoparticles. The TEM images showed that the γ-Fe2O3 and Fe3O4 were spherical, and their size was 18 and 22 nm respectively. Magnetic properties have been measured by VSM at room temperature. Hysteresis loops showed that the γ-Fe2O3 and Fe3O4 nanoparticles were super-paramagnetic.
Similar content being viewed by others
References
Jing S., Zhou S., Hou P., Yang Y., Weng J., Li X., Li M., J. Biomed. Mater. Res., A 80 (2007), 333.
Wang X., Zhang R., Wu C., Dai Y., Song M., Gutmann S., Gao F., Lv G., Li J., Li X., Guan Z., Fu D., Chen B., J. Biomed. Mater. Res., A 80 (2007), 852.
Yuksel K., J. Magn. Mater., 300 (2006), 327.
Iida H., Takayanagi K., Nakanishi T., Osaka T., J. Colloid Interface Sci., 314 (2007), 274.
Ling H.L., Pilko S., Wu J.H., Jung M.H., Min J.H., Lee J.H., An B.H., Kim Y.K., J. Magn. Mater., 310 (2007), 815.
Lian S., Kang Z., Wang E., Jiang M., Hu C., Solid State Commun., 127(2003), 605.
Sadeghi M., Sarabadani P., Karami H., J. Magn. Mater., 283 (2010), 297.
Abareshi M., Goharshadi E.K., Zebarjad S.M., Khandan Fadafan H., Yussefi A., J. Magn. Mater., 322 (2010), 3895.
Shen Y.F., Tang J., Nie Z.H., Wang Y.D., Ren Y., Zuo L., J. Separ. Purif. Technol., 68 (2009), 312.
Darezereshki E., J. Mater. Lett., 64 (2010), 1471.
Darezereshki E., J. Mater. Lett., 65 (2010), 642.
Sellmyer D., Skomski R., Advanced magnetic nanostructures, Springer, New York, 2006.
Dutz S., Hergt R., J. Nano-Electron. Phys., 4 (2012), 02010.
Alibeigi S., Vaezi M.R., J. Chem. Eng. Technol., 31 (2008), 1591.
Mody V.V., Siwale R., Singh A., Mody H.R., Adv. Powder. Technol., 2 (2010), 282.
Lai J., Shafi K.V.P.M., Loos K., Ulnam A., Lee Y., Vogt T., Estornes C., J. Structure. AM. Chem. Soc., 125 (2003), 11470.
Xu X.N., Wolfus Y., Shaulov A., Yeshurun Y., J. App. Phys., 91 (2002), 4611.
Cornell R.M., Schwertmann U., The iron oxides: structure, properties, reactions, occurrences. JohnWiley, Cambridge, 2003.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Aliahmad, M., Nasiri Moghaddam, N. Synthesis of maghemite (γ-Fe2O3) nanoparticles by thermal-decomposition of magnetite (Fe3O4) nanoparticles. Mater Sci-Pol 31, 264–268 (2013). https://doi.org/10.2478/s13536-012-0100-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s13536-012-0100-6