Abstract
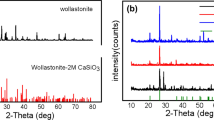
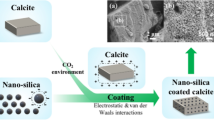
Nano calcium carbonate particles have a wide range of industrial applications due to their beneficial properties such as high porosity and high surface area to volume ratio and due to their strengthening the mechanical properties of plastics and paper. Consequently, significant research has been done to deliver a new approach for the synthesis of precipitated nano calcium carbonate by using a carbonation process through a closed loop reactor. Both the experimental and the instrumental parameters, i.e. the CO2 flow rate, the concentration of the starting materials (Ca(OH)2 and CaO), the pH, the orifice diameter, etc., were investigated. The carbonation efficiency was increased due to the diffusion process involved in the loop reactor. The particle size was affected by the CO2 flow rate, reaction time, and orifice diameter. Finally, precipitated nano calcite calcium carbonate (50 to 100 nm) was synthesized by optimizing all the experimental and the instrumental parameters. The synthesized precipitated nano calcium carbonate was characterized by using scanning electron microscopy (SEM), X-ray diffraction (XRD), and Fourier transform infrared (FTIR) spectroscopy. This study has proved that the carbonation efficiency can be enhanced for a short time by using a loop reactor and that the carbonation process was more energy efficient and cost effective than other conventional methods.
Similar content being viewed by others
References
C. Y. Tai and F. B. Chen, AIChE J. 44, 1790 (1998).
H. Casanova and L. P. Higuita, J. Chem. Eng. 175, 569 (2011).
M. Huber, W. J. Stark, S. Loher, M. Maciejewski, F. Krumeich and A. Baiker, Chem. Commun. 5, 648 (2005).
K. Eguchil, T. Satake, M. Tajika, S. Kuwamoto, K. Yokoyama, J. Matsui, T. Issiki and N. mesaki, IOP Conf. Series: Mat. Sci. Eng. 18, 062001 (2011).
T. Tsuzuki, K. Pethick and P. G. McCormick, J. Nanopart. Res. 2, 375 (2000).
A. Cai, X. Xu, H. Pan, J. Tao, R. Liu, R. Tang and K. Cho, J. Phys. Chem. 112, 11324 (2008).
J. F. Chen, Y.-H. Wang, F. Guo, X.-M. Wang and C. Zheng, Ind. Eng. Chem. Res. 39, 948 (2000).
A. G. J. Ghadam, M. Idrees and S. A. H. Naqvi, Intern. J. Adv. Engin. Sci. 2, 1 (2012).
G. Montes-Hernandez, A. Fernandez-Martinez, L. Charlet, D. Tisserand and F. Renard, J. Cryst. Growth 310, 2946 (2008).
C. Y. Ma and X. Z. Wang, J. Process Control 22, 72 (2012).
Y. Yang, C. Y. Ma and X. Z. Wang, The Int. Federation of Automatic Control (2012), p. 216.
I. Nishida, Ultrasonics Sonochem. 11, 423 (2004).
S. H. Sonawane, S. P. Gumfekar, K. H. Kate, S. P. Meshram, K. J. Kunte, L. Ramjee, C. M. Mahajan, M. G. Parande and M. Ashokkumar, Intern. J. Chem. Engin. 2010, 1 (2010).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thriveni, T., Ahn, J.W., Ramakrishna, C. et al. Synthesis of nano precipitated calcium carbonate by using a carbonation process through a closed loop reactor. Journal of the Korean Physical Society 68, 131–137 (2016). https://doi.org/10.3938/jkps.68.131
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3938/jkps.68.131